Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Zhejiang Yixin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.herbs-tech.com | |||
 | +86 (579) 8886-5135 8886-5136 8886-5105 | |||
 | +86 (579) 8886-5169 | |||
 | sale@herbs-tech.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Bio-sugars Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bio-sugars.com | |||
 | +86 (579) 8901-8388 +86 15167971485 | |||
 | +86 (579) 8855-6787 | |||
 | syngars@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Beijing Ribio Biotech. Inc. | China | |||
|---|---|---|---|---|
 | www.ribio.com.cn | |||
 | +86 (10) 6266-4360 +86 13910960639 | |||
 | +86 (10) 6266-4370 | |||
 | sales@ribio.com.cn ribio@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| SL Drugs and Pharmaceuticals Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.sldrugs.com | |||
 | +91 (40) 6661-1133 | |||
 | +91 (40) 2375-1130 | |||
 | enquiry@sldrugs.com | |||
| Chemical distributor since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhejiang Synose Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.synose.com | |||
 | +86(579)8227-5537 8917-2081 | |||
 | +86 (579) 8227-7133 | |||
 | expo@synose.com | |||
| Chemical manufacturer since 1996 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Chiralblock Bioscience LLC | China | |||
|---|---|---|---|---|
 | www.chiralblock.com | |||
 | +86 18143482572 | |||
 | +86 (571) 8848-7672 | |||
 | info@chiralblock.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2012 | ||||
| Venkateshwara Life Sciences | India | |||
|---|---|---|---|---|
 | www.venkateshwaralifesciences.com | |||
 | +91 (9963) 894-542 | |||
 | balakrishna@venkateshwaralifesciences.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Creative Proteomics | USA | |||
|---|---|---|---|---|
 | www.creative-proteomics.com | |||
 | +1 (631) 593-0501 | |||
 | +1 (631) 614-7828 | |||
 | contact@creative-proteomics.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Xinxiang Aurora Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aurora-biotech.com | |||
 | +86 (373) 308-8722 +86 18637352520 | |||
 | +86 (373) 308-8722 | |||
 | info@aurora-biotech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Jiangsu United-value International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.uvcs.cn.com | |||
 | +86 (519) 8988-5611 | |||
 | +86 (519) 8988-5615 | |||
 | luke.li@uvcs.cn.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Zhejiang Medicines & Health Products Import & Export Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zmcchina.com | |||
 | +86 (0571) 281-9008 | |||
 | anna@zmcchina.com | |||
| Chemical distributor since 1984 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Ramidus AB | Sweden | |||
|---|---|---|---|---|
 | www.ramidus.se | |||
 | +46 (46) 286-2850 | |||
 | +46 (46) 286-2851 | |||
 | info@ramidus.se | |||
| Chemical manufacturer | ||||
| Orgentis Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.orgentis.com | |||
 | +49 39482-355 | |||
 | +49 39482-79071 | |||
 | sales@orgentis.com | |||
| Chemical manufacturer since 1991 | ||||
| Xinxiang Tianfeng Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianfengchem.com | |||
 | +86 (373) 251-3858 | |||
 | +86 (373) 251-3700 | |||
 | tianfeng@tianfengchem.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Carbohydrate >> Monosaccharide |
|---|---|
| Name | Diacetone-D-glucose |
| Synonyms | 1,2:5,6-Diisopropylidene-D-glucose; 1,2,5,6-Diisopropylidene-D-glucose; D-Glucose diacetonide; Diacetone glucose |
| Molecular Structure | 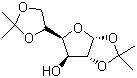 |
| Molecular Formula | C12H20O6 |
| Molecular Weight | 260.29 |
| CAS Registry Number | 582-52-5 |
| EC Number | 209-486-0 |
| SMILES | CC1(OCC(O1)[C@@H]2[C@@H]([C@@H]3[C@H](O2)OC(O3)(C)C)O)C |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 110 - 111 °C (Expl.) |
| Boiling point | 362.8±42.0 °C 760 mmHg (Calc.)* |
| Flash point | 173.2±27.9 °C (Calc.)* |
| Index of refraction | 1.483 (Calc.)* |
| alpha | -11 ° (c=5, EtOH) |
| Water solubility | slightly soluble |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Diacetone-D-glucose is a protected derivative of D-glucose in which two pairs of hydroxyl groups are converted into isopropylidene acetals through reaction with acetone. D-glucose was identified in the nineteenth century as a fundamental carbohydrate present in fruits, honey, and as a product of starch hydrolysis. Its cyclic pyranose structure and stereochemical configuration were established through classical chemical studies, including mutarotation experiments and formation of crystalline derivatives. As carbohydrate chemistry advanced in the early twentieth century, the development of reliable protecting group strategies became essential for selective modification of the multiple hydroxyl groups present in monosaccharides. The reaction of sugars with acetone in the presence of an acid catalyst was shown to produce cyclic acetals, commonly referred to as acetonides. In the case of D-glucose, treatment with acetone under controlled acidic conditions yields diacetone-D-glucose, in which two isopropylidene groups are introduced. The most widely described form is 1,2:5,6-di-O-isopropylidene-D-glucose, where the hydroxyl groups at positions 1 and 2 form one acetonide ring and those at positions 5 and 6 form a second. This transformation was documented as part of systematic investigations into carbohydrate protection and provided a stable, crystalline derivative suitable for further chemical manipulation. The discovery and characterization of diacetone-D-glucose contributed significantly to the refinement of synthetic carbohydrate methodology. By masking four of the five hydroxyl groups of glucose, chemists were able to control reactivity and direct transformations to the remaining free hydroxyl group. The reduction of intermolecular hydrogen bonding in the protected derivative increases solubility in organic solvents such as acetone, ether, and chloroform, facilitating purification and reaction under anhydrous conditions. These properties made diacetone-D-glucose a practical intermediate in laboratory synthesis. Diacetone-D-glucose has been used extensively as a starting material for the preparation of selectively substituted glucose derivatives. The free hydroxyl group can undergo reactions such as esterification, ether formation, oxidation, or substitution without interference from the protected positions. After the desired modification is achieved, the isopropylidene groups can be removed by treatment with dilute acid, regenerating the corresponding diol functionalities. The predictable formation and cleavage of acetonide protecting groups have been documented in numerous studies and form a cornerstone of carbohydrate protecting group chemistry. Beyond its role in synthetic methodology, diacetone-D-glucose has been employed in investigations of reaction mechanisms and stereochemical effects in carbohydrate chemistry. The rigidifying influence of the acetonide rings can affect conformational preferences, and comparisons between protected and unprotected glucose derivatives have provided insight into structural behavior. Experimental observations derived from such studies have informed broader understanding of hexose chemistry. Although diacetone-D-glucose itself is primarily used as an intermediate rather than a final consumer product, its preparation and application are well established in the literature. The compound exemplifies the practical implementation of protecting group strategies that enabled systematic synthesis of complex carbohydrate derivatives. Through its documented role in controlled functionalization and structural investigation, diacetone-D-glucose represents an important development in the experimentally verified evolution of modern glycochemistry. References 2021. Site-specific incorporation of 5′-methyl DNA enhances the therapeutic profile of gapmer ASOs. Nucleic Acids Research. DOI: 10.1093/nar/gkab047 2010. Efficient synthesis of 6-O-palmitoyl-1,2-O-isopropylidene-α-d-glucofuranose in an organic solvent system by lipase-catalyzed esterification. Biotechnology Letters. DOI: 10.1007/s10529-010-0334-5 |
| Market Analysis Reports |