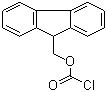
9-Fluorenylmethyl chloroformate (FMOC-Cl) is a versatile chemical reagent that is widely used in organic synthesis and analytical chemistry, especially peptide synthesis. Its introduction has significantly improved the efficiency and specificity of chemical processes in these fields.
FMOC-Cl was originally developed in the 1970s as part of an effort to improve peptide synthesis methods. The goal was to create a reagent that could protect amino groups during synthesis, preventing unwanted side reactions and allowing for more precise construction of peptide chains. The usefulness of FMOC-Cl stems from its ability to form stable but easily removable protecting groups on amino acids, called FMOC groups.
One of the main applications of FMOC-Cl is solid phase peptide synthesis (SPPS). In this process, amino acids are sequentially added to a growing peptide chain that is fixed to an insoluble resin. The amino group of each amino acid is protected by a FMOC group, which prevents it from reacting prematurely. Once the amino acid is attached to the peptide chain, the FMOC group can be selectively removed using a weak base such as piperidine, exposing the amino group for the next coupling reaction. This cycle of protection, coupling, and deprotection allows for efficient and precise peptide assembly.
FMOC-Cl offers several advantages over other protecting groups used in peptide synthesis. It is stable under the acidic conditions typically used to remove peptides from resins, but can be easily removed under mildly basic conditions. This selectivity reduces side reactions and increases the yield and purity of the final peptide product. Additionally, the relatively large size of the FMOC group provides steric hindrance, which helps minimize undesirable reactions during synthesis.
In addition to peptide synthesis, FMOC-Cl is used for amino acid derivatization for high-performance liquid chromatography (HPLC) analysis. Amino acids are derivatized with FMOC-Cl to form FMOC-amino acid derivatives, which are more easily detected and separated by HPLC. This application is critical for the quantitative analysis of amino acids in biological samples, foods, and other complex mixtures.
In addition to its role in peptide synthesis and amino acid analysis, FMOC-Cl is used in the preparation of other organic compounds. It protects amino groups during a variety of synthetic processes, allowing chemists to control the reactivity of versatile molecules. This makes FMOC-Cl a valuable tool in pharmaceutical synthesis, where precise control of molecular structure is critical.
The discovery of FMOC-Cl has also led to advances in materials science. For example, FMOC-protected amino acids can be used to create peptide-based nanomaterials with specific properties and functions. These materials can be used in drug delivery, tissue engineering, and scaffolds for cell growth. The ability to precisely control peptide sequence and structure using FMOC-Cl is essential for developing these advanced materials.
In biochemical research, FMOC-Cl is used to study protein interactions and function. By synthesizing peptides with specific sequences and modifications, researchers can study how proteins interact with each other and with other molecules. This information is essential for understanding cellular processes and developing new therapeutic strategies.
Although FMOC-Cl is widely used, it requires caution when handling. It is an active compound that may irritate the skin, eyes, and respiratory system. Appropriate safety measures must be taken when working with FMOC-Cl, including the use of personal protective equipment and adequate ventilation.
References
2024. Possible fetal bovine serum-reduced boost cultivation of DF-1 by supplementation with plant peptones and yeast extracts. Food Science and Biotechnology.
DOI: 10.1007/s10068-024-01774-y
2024. A facile optical sensing strategy for glyphosate detection based on structure-switching signaling aptamers. Microchimica Acta.
DOI: 10.1007/s00604-024-06839-7
2024. Effects of High-Pressure Processing on the Biosynthesis of γ-Aminobutyric Acid in Cocoa Beans Fermented with Lactobacillus senmaizukei. Food and Bioprocess Technology.
DOI: 10.1007/s11947-024-03651-2
|









































































 GHS06;GHS07 Danger Details
GHS06;GHS07 Danger Details