Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Sichuan Zhongbang Technical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhongbangst.com | |||
 | +86 (830) 258-5019 +86 18982761636 | |||
 | +86 (830) 258-9033 | |||
 | sales@zhongbangst.com sheldon0chen@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Qyubiotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qyubiotech.com | |||
 | +86 (021) 3759-6619 | |||
 | +86 (021) 3759-6619 | |||
 | lioner@qyubiotech.com | |||
 | QQ Chat | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Endotherm GmbH | Germany | |||
|---|---|---|---|---|
 | www.endotherm-lsm.com | |||
 | +49 (681) 3946-7570 | |||
 | +49 (681) 3946-7571 | |||
 | info@endotherm.de | |||
| Chemical manufacturer | ||||
| Classification | Natural product >> Natural phenols |
|---|---|
| Name | 4-(1-Adamantyl)phenol |
| Synonyms | 4-(Adamantan-1-yl)phenol |
| Molecular Structure | 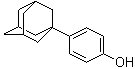 |
| Molecular Formula | C16H20O |
| Molecular Weight | 228.33 |
| CAS Registry Number | 29799-07-3 |
| EC Number | 631-106-0 |
| SMILES | C1C2CC3CC1CC(C2)(C3)C4=CC=C(C=C4)O |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 181-183 °C (Expl.) |
| Index of Refraction | 1.612, Calc.* |
| Boiling Point | 361.6±21.0 °C (760 mmHg), Calc.* |
| Flash Point | 190.3±11.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  Xi Details Xi Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | R36/37/38 Details | ||||||||||||||||
| Safety Statements | S26;S36 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
4-(1-Adamantyl)phenol, also known as adamantylphenol, is an organic compound that consists of a phenol molecule with an adamantyl group attached at the para-position of the benzene ring. The chemical formula for this compound is C15H20O, and it is characterized by the presence of a phenol structure (C6H5OH) and an adamantyl group (C10H15), which is a bicyclic hydrocarbon derived from adamantane. The compound was first synthesized as part of a broader effort to explore the chemistry of adamantane derivatives. Adamantane itself was first isolated in the 1930s, and since then, many derivatives of this bicyclic structure have been synthesized for various applications. The synthesis of 4-(1-adamantyl)phenol involves the reaction of adamantyl compounds with phenol under suitable conditions, introducing the adamantyl group to the phenolic ring at the para position. 4-(1-Adamantyl)phenol has several notable applications in organic chemistry and materials science. One of its primary uses is as an intermediate in the synthesis of more complex compounds, particularly in the field of pharmaceuticals and agrochemicals. Its structure makes it a useful building block in the development of compounds with unique physical and chemical properties, including those with potential biological activity. In addition, 4-(1-adamantyl)phenol has been explored for its potential use as a stabilizer in various chemical reactions. The adamantyl group, being a bulky and rigid structure, can provide steric protection to the phenolic group, enhancing the stability of the compound in certain chemical environments. This property is of interest in the design of stable materials or molecules that need to withstand harsh conditions without undergoing degradation. The compound has also been studied for its potential applications in the field of catalysis. It can be used as a ligand in coordination chemistry, where the adamantyl group may influence the coordination environment and reactivity of metal centers. This makes 4-(1-adamantyl)phenol a candidate for use in catalysis processes, particularly in reactions requiring the stabilization of metal ions. Moreover, 4-(1-adamantyl)phenol has been researched for its potential biological activities. Although it has not been widely used as a therapeutic agent, studies have suggested that adamantyl derivatives, including 4-(1-adamantyl)phenol, could exhibit antimicrobial, antiviral, or anticancer properties. The specific mechanisms of action and efficacy of such compounds are still under investigation, and the compound's role in these areas remains a subject of ongoing research. In conclusion, 4-(1-adamantyl)phenol is a versatile organic compound with applications primarily in the fields of organic synthesis, materials science, and catalysis. Its unique structure, combining the rigid adamantyl group with the reactive phenol group, provides opportunities for further exploration in the development of new chemical materials and bioactive compounds. As research continues, the potential uses of 4-(1-adamantyl)phenol in both industrial and biomedical applications may expand. References 2009. Exploration of endocrine-disrupting chemicals on estrogen receptor alpha by the agonist/antagonist differential-docking screening (AADS) method: 4-(1-adamantyl)phenol as a potent endocrine disruptor candidate. Toxicology Letters, 191(2-3). DOI: 10.1016/j.toxlet.2009.08.001 2018. Theory and Practice of the Preparation of Adamantylarenes. Petroleum Chemistry, 58(10). DOI: 10.1134/s0965544118100171 2020. Thermal Stability Study of 4-(1-Adamantyl)phenol. Petroleum Chemistry, 60(11). DOI: 10.1134/s0965544120110171 |
| Market Analysis Reports |