Online Database of Chemicals from Around the World
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hubei Chemworth Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemworth.com | |||
 | +86 (27) 8423-5352 +86 13971178775 | |||
 | +86 (27) 8423-5352 | |||
 | sales@chemworth.com sinon_wangwei@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty acid |
|---|---|
| Name | N-Phenylbenzohydroxamic acid |
| Synonyms | N-Benzoyl-N-phenylhydroxylamine; N-Hydroxy-N-phenylbenzamide |
| Molecular Structure | 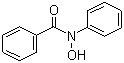 |
| Molecular Formula | C13H11NO2 |
| Molecular Weight | 213.23 |
| CAS Registry Number | 304-88-1 |
| EC Number | 206-158-9 |
| SMILES | C1=CC=C(C=C1)C(=O)N(C2=CC=CC=C2)O |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 118 - 120 $degree$C (Expl.) |
| Boiling point | 369.2$+/-$25.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 177.1$+/-$23.2 $degree$C (Calc.)* |
| Index of refraction | 1.666 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
N-Phenylbenzohydroxamic acid is an aromatic hydroxamic acid derivative containing both benzoyl and phenyl substituents attached to a hydroxamic acid functional group. Its molecular formula is C13H11NO2. The compound belongs to the class of hydroxamic acids, which are characterized by the functional group R–C(=O)–N(OH)–R′ and are widely studied for their metal-chelating properties and synthetic utility. Structurally, N-phenylbenzohydroxamic acid consists of a benzoyl group attached to a hydroxamic acid nitrogen atom that also bears a phenyl substituent. The molecule therefore contains two aromatic rings connected through a conjugated carbonyl–N-hydroxylamine linkage. The hydroxamic acid moiety includes both a carbonyl group and an N-hydroxyl group, enabling resonance stabilization and tautomeric behavior. The hydroxamic acid functional group is the defining chemical feature of the compound. Electron delocalization occurs across the carbonyl, nitrogen, and hydroxyl oxygen atoms, giving rise to partial double-bond character in the C–N bond and contributing to planarity of the functional group. The compound can exist in different tautomeric forms involving proton transfer between the oxygen and nitrogen atoms. One of the most important properties of hydroxamic acids is their ability to chelate metal ions. N-Phenylbenzohydroxamic acid can coordinate to metal centers through the carbonyl oxygen and the deprotonated hydroxyl oxygen, forming stable five-membered chelate rings. This metal-binding behavior has made hydroxamic acid derivatives important ligands in coordination chemistry and analytical chemistry. Hydroxamic acids have historically attracted considerable attention because of their occurrence in biologically active compounds and their ability to inhibit metalloproteins. The strong affinity of hydroxamate groups for metal ions, especially iron and zinc, underlies many of their biochemical interactions. N-Phenylbenzohydroxamic acid has been used in coordination chemistry studies to investigate complex formation with transition metals. The aromatic substituents influence both steric and electronic properties of the ligand, affecting complex stability and geometry. Such compounds have also been investigated as analytical reagents for selective metal ion extraction and detection. The compound can be synthesized through reaction of benzoyl chloride or related activated benzoic acid derivatives with N-phenylhydroxylamine. In this acylation reaction, the nucleophilic hydroxylamine derivative attacks the activated carbonyl carbon, forming the hydroxamic acid linkage after elimination of the leaving group. Chemically, N-phenylbenzohydroxamic acid can undergo reactions typical of hydroxamic acids, including hydrolysis, metal complexation, oxidation, and acyl transfer reactions. Under strongly acidic or basic conditions, hydrolysis may cleave the hydroxamic acid bond to yield benzoic acid derivatives and substituted hydroxylamines. From a physicochemical standpoint, the molecule exhibits moderate polarity because of the hydroxamic acid functionality, while the two aromatic rings contribute substantial hydrophobic character. The hydroxyl and carbonyl groups participate in hydrogen bonding, influencing crystal packing and solubility behavior. The conjugated aromatic system and resonance-stabilized hydroxamic acid group also contribute to characteristic ultraviolet absorption properties. The compound may exist in different conformations depending on hydrogen bonding and solvent interactions. Historically, hydroxamic acids became increasingly important in the development of coordination chemistry and medicinal chemistry during the 20th century. Studies of aromatic hydroxamic acids such as N-phenylbenzohydroxamic acid contributed to understanding the metal-chelating behavior and electronic structure of this functional group. Overall, N-phenylbenzohydroxamic acid is an aromatic hydroxamic acid derivative characterized by a benzoyl-linked N-hydroxylamine structure bearing two phenyl groups. Its resonance-stabilized hydroxamic acid functionality gives it strong metal-chelating ability and important reactivity in coordination chemistry, analytical chemistry, and synthetic organic chemistry. References 2026. Separation of Niobium and Tantalum Contained in Tin Slag Leach Liquor Using a Hydroxypropyl-picolylamine Functional Group Ion Exchange Chelating Resin. Journal of Sustainable Metallurgy. DOI: 10.1007/s40831-025-01382-y 2025. Separation of no-carrier added 90Nb from proton irradiated bulk zirconium target using curcumin. Journal of Radioanalytical and Nuclear Chemistry. DOI: 10.1007/s10967-025-10415-5 |
| Market Analysis Reports |