Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Changzhou Sunchem Pharmaceutical Chemical Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunkechem.com | |||
 | +86 (519) 8519-5575 8519-5528 8519-5565 | |||
 | +86 (519) 8519-5585 | |||
 | info@sunkechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jinan Dechenghemu Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jndcyy.com | |||
 | +86 (531) 8888-3739 | |||
 | +86 (531) 6232-5949 | |||
 | dcyy@vip.sina.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Suzhou No.5 Pharmaceutical Factory Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.suzhoupharm.com | |||
 | +86 (512) 6535-2055 | |||
 | +86 (512) 6535-9935 | |||
 | szwy@public1.sz.js.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hubei Yitai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbyitai.com.cn | |||
 | +86 (27) 8588-2668 | |||
 | +86 (27) 8588-2668 | |||
 | elsie@hbyitai.com.cn | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Creative Proteomics | USA | |||
|---|---|---|---|---|
 | www.creative-proteomics.com | |||
 | +1 (631) 593-0501 | |||
 | +1 (631) 614-7828 | |||
 | contact@creative-proteomics.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Chongqing Huapont Pharm. Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huapont.com.cn | |||
 | +86 (23) 6788-9222 6788-6928 | |||
 | +86 (23) 6788-9222 / 6788-6927 | |||
 | huapont@huapont.com.cn | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Biochemical >> Carbohydrate >> Monosaccharide |
|---|---|
| Name | D(+)-Glucurono-3,6-lactone |
| Synonyms | D-Glucurone |
| Molecular Structure | 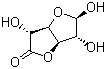 |
| Molecular Formula | C6H8O6 |
| Molecular Weight | 176.12 |
| CAS Registry Number | 32449-92-6 |
| EC Number | 251-053-3 |
| SMILES | C(=O)[C@@H]([C@@H]1[C@@H]([C@@H](C(=O)O1)O)O)O |
| Density | 1.8±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 170-176 °C (Expl.) |
| Boiling point | 403.5±28.0 °C 760 mmHg (Calc.)* |
| Flash point | 174.9±17.5 °C (Calc.)* |
| Solubility | water soluble (Expl.) |
| Index of refraction | 1.597 (Calc.)*, 19 ° (c=10, H2O) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||||||||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
D(+)-Glucurono-3,6-lactone, more commonly known as glucuronolactone (glucurolactone; D-glucuronolactone), is a cyclic ester (lactone) derivative of D-glucuronic acid. Its structure involves internal esterification between the carboxyl group and a hydroxyl group, forming a ring that makes it more chemically stable than the free acid. In human and animal metabolism, glucuronolactone is an intermediate within the ascorbate and aldarate metabolic pathways, formed by reduction of L-gulono-1,4-lactone and subject to enzymatic lactonization and hydrolysis reactions via uronolactonase and related enzymes. The historical “discovery” of glucuronolactone traces back to studies in carbohydrate and vitamin C metabolism during the mid-20th century. Researchers investigating the biosynthesis of ascorbic acid and sugar acid interconversions identified glucuronolactone among endogenous metabolites in animals and plants. Over time its presence was confirmed in connective tissues, blood plasma, and plant gums, as well as in dietary sources. In practical applications, glucuronolactone has seen use mostly as a supplemental ingredient in energy drinks and nutritional formulations. It is commonly added under the assumption that it supports detoxification pathways—particularly glucuronidation in the liver—and enhances recovery or cognitive performance, though scientific support for such claims is limited. ([Wikipedia][2]) Some regulatory and safety assessments, such as by EFSA, have examined its use in combination with caffeine and taurine, concluding that current evidence does not indicate harmful interactions at typical consumption levels. ([Wikipedia][2]) Metabolically, glucuronolactone plays a role in uronic acid pathways: it can be hydrolyzed to glucuronate (D-glucuronate), which then participates in conjugation reactions (for example, glucuronidation of xenobiotics and endogenous molecules), enhancing their solubility and facilitating excretion. Enzymes such as uronolactonase catalyze this hydrolysis. ([Wikipedia][3]) In some species, the interconversion of glucuronolactone and related lactones is tightly linked with ascorbic acid (vitamin C) biosynthesis pathways. ([Wikipedia][1]) Aside from its role in supplemental formulations, glucuronolactone has been utilized in studies of connective tissue metabolism, where it is considered a structural component of glycosaminoglycans and polysaccharides. Its presence in plant gums and exudates suggests a structural or protective function in plants. ([Wikipedia][2]) Analytical and biochemical research sometimes employs glucuronolactone or its derivatives as standards in metabolic profiling, especially in studying sugar acid pathways or detoxification enzyme activities. ([hmdb.ca][4]) In summary, D(+)-glucurono-3,6-lactone (glucuronolactone) is a naturally occurring metabolite in carbohydrate and detoxification biochemistry. It was identified early in studies of sugar acid metabolism and ascorbate biosynthesis, and it is applied today primarily in nutritional and energy drink formulations with claims related to detoxification or recovery. While its endogenous metabolic roles are reasonably well characterized, its efficacy and utility in supplementation remain areas of ongoing inquiry. References 2013. Retinol and retinyl esters: biochemistry and physiology. Journal of Lipid Research, 54(7). DOI: [https://doi.org/10.1194/jlr.R037648](https://doi.org/10.1194/jlr.R037648) 2009. Speciation of selenium dietary supplements; formation of Se-methylselenocysteine and related species. Analytica Chimica Acta, 652(1-2). DOI: [https://doi.org/10.1016/j.aca.2009.08.013](https://doi.org/10.1016/j.aca.2009.08.013) |
| Market Analysis Reports |