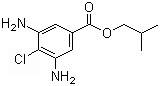
Isobutyl 3,5-diamino-4-chloro benzoate is an organic compound with significant applications in various chemical and pharmaceutical industries. This compound belongs to the class of benzoates, which are esters of benzoic acid. It is characterized by the presence of an isobutyl group at the ester position, along with two amino groups and a chloro substituent at specific positions on the benzene ring. These functional groups contribute to its reactivity and its potential for use in diverse chemical processes.
The discovery of Isobutyl 3,5-diamino-4-chloro benzoate is linked to ongoing research in the field of organic chemistry, particularly in the synthesis of compounds with functionalized aromatic rings. The benzoate esters, including this compound, are typically synthesized by esterification reactions between benzoic acid derivatives and alcohols. The amino and chloro groups on the benzene ring are introduced through selective halogenation and amination reactions. These functional groups significantly affect the compound's properties, making it a valuable intermediate in organic synthesis.
The primary applications of Isobutyl 3,5-diamino-4-chloro benzoate are seen in the pharmaceutical industry, where it serves as an intermediate in the synthesis of more complex molecules. It has been used in the development of specific active pharmaceutical ingredients (APIs) due to its ability to undergo further chemical transformations, such as nucleophilic substitution and condensation reactions. The amino groups in particular make it useful for the synthesis of compounds with enhanced biological activity. The presence of the chloro group also contributes to its reactivity, allowing for further modification and functionalization, which is essential in the development of novel therapeutic agents.
In addition to its pharmaceutical uses, Isobutyl 3,5-diamino-4-chloro benzoate has found applications in agrochemical formulations. It has been studied for its potential in the synthesis of herbicides, fungicides, and other crop protection agents. The structure of the compound, with its combination of functional groups, offers possibilities for designing molecules that can interact effectively with biological systems, including enzymes and receptors involved in plant growth and pathogen resistance.
The compound’s utility extends into the field of materials science, where it is explored for its potential in the production of novel polymers and coatings. The chemical structure of Isobutyl 3,5-diamino-4-chloro benzoate can contribute to the development of materials with specific properties, such as improved chemical resistance or enhanced mechanical strength, due to its unique combination of functional groups.
In summary, Isobutyl 3,5-diamino-4-chloro benzoate is a versatile chemical intermediate with applications spanning pharmaceuticals, agrochemicals, and materials science. Its discovery has been instrumental in advancing the synthesis of complex molecules and the development of functionalized materials, offering valuable contributions to various industrial fields.
References
2013. Synthesis and chemical transformations of 6-amino-7-chloro-2-(2-hydroxyphenyl)-2H-benzo-triazole-4-carboxylic acid derivatives. Chemistry of Heterocyclic Compounds, 48(10).
DOI: 10.1007/s10593-013-1165-9
|




























 GHS07 Warning Details
GHS07 Warning Details