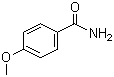
4-Methoxybenzamide is an aromatic amide compound with the molecular formula C7H9NO2. It consists of a benzene ring substituted with a methoxy group (-OCH3) at the para position and an amide group (-CONH2). This compound is part of a larger class of substituted aromatic amides, which are widely used in various fields of chemical research and application due to their versatile chemical properties. The discovery and development of 4-methoxybenzamide can be traced to advancements in organic chemistry focused on the functionalization of aromatic compounds, especially with the introduction of substituent groups that can modify their reactivity and enhance their applications in different industries.
The synthesis of 4-methoxybenzamide typically involves straightforward methods such as the acylation of 4-methoxyaniline with acylating agents, like acyl chlorides, to introduce the amide functional group. This reaction forms the amide bond between the 4-methoxyphenyl group and the carbonyl group, resulting in the formation of 4-methoxybenzamide. The methoxy group, being an electron-donating group, stabilizes the aromatic ring and influences the reactivity of the compound, making it a useful intermediate in various synthetic processes.
One of the major applications of 4-methoxybenzamide lies in the pharmaceutical industry. The compound's amide functionality and aromatic structure make it a valuable scaffold for the design and synthesis of bioactive molecules. In particular, 4-methoxybenzamide derivatives have been explored for their potential as anti-inflammatory, antimicrobial, and anticancer agents. The methoxy group in the para position of the aromatic ring can significantly impact the compound's interaction with biological targets, often enhancing the compound's bioactivity and influencing its pharmacokinetic properties. In some studies, 4-methoxybenzamide and its derivatives have been shown to exhibit promising activities in modulating enzyme activity, which is crucial for the development of new therapeutic agents.
In addition to its pharmaceutical uses, 4-methoxybenzamide is also valuable in the field of material science. Aromatic amides are known for their excellent thermal stability and are often used as precursors in the synthesis of high-performance polymers. 4-Methoxybenzamide, due to the presence of the methoxy group, is a suitable candidate for creating polymers with specific electronic properties. The compound's stable and reactive structure makes it useful in the production of engineering plastics, coatings, and films. The introduction of the methoxy group, in particular, can enhance the solubility and processability of these materials, which is essential for their application in advanced material technologies.
The compound is also employed in synthetic organic chemistry as a starting material or intermediate for the synthesis of more complex organic molecules. Its well-defined structure allows it to be used in various reaction schemes, including the preparation of other functionalized aromatic compounds. These compounds are important for the development of a wide range of chemical products, including agrochemicals, fragrances, and specialty chemicals.
Moreover, 4-methoxybenzamide has been utilized in the synthesis of various organic ligands used in coordination chemistry. These ligands are key in the preparation of metal complexes, which have diverse applications in catalysis, material science, and medicinal chemistry. The ability of 4-methoxybenzamide to coordinate with metals and form stable complexes makes it an essential tool in the development of new catalysts and in the study of metal-ligand interactions.
In conclusion, 4-methoxybenzamide is a versatile compound with a broad range of applications in pharmaceutical, material science, and synthetic chemistry. Its synthesis and functionalization open doors for creating bioactive molecules, high-performance materials, and new catalysts. The discovery and continued exploration of 4-methoxybenzamide offer numerous possibilities for advancing research and technology across various scientific disciplines.
References
2010. Recent Advances in the Synthesis of Amides. Tetrahedron, 66(6).
DOI: 10.1016/j.tet.2009.11.090
|





































 GHS07 Warning Details
GHS07 Warning Details