Online Database of Chemicals from Around the World
| Novasyn Organics Pvt. Ltd. | India | |||
|---|---|---|---|---|
 | www.novasynorganics.com | |||
 | +91 (406) 620-2233 633-9111 | |||
 | +91 (402) 781-1997 | |||
 | info@novasynorganics.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Fred Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fredbio.com | |||
 | +86 (25) 8469-6168 | |||
 | Austin@fredchem.cn | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: NJFred01 | |||
 | WhatsApp:+86 17302533743 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Maybridge Chemical Co., Ltd. | Belgium | |||
|---|---|---|---|---|
 | www.maybridge.com | |||
 | +32 (14) 593-434 | |||
 | maybridge.sales@thermofisher.com | |||
| Chemical manufacturer | ||||
| AOBChem USA | USA | |||
|---|---|---|---|---|
 | www.aobchem.com | |||
 | +1 (323) 382-7678 | |||
 | +1 (213) 687-8698 | |||
 | sales@aobchem.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Pyridine derivative |
|---|---|
| Name | 5-Bromonicotinonitrile |
| Synonyms | 5-Bromo-3-cyanopyridine; 5-Bromopyridine-3-carbonitrile |
| Molecular Structure | 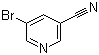 |
| Molecular Formula | C6H3BrN2 |
| Molecular Weight | 183.00 |
| CAS Registry Number | 35590-37-5 |
| EC Number | 625-836-9 |
| SMILES | C1=C(C=NC=C1Br)C#N |
| Density | 1.7±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 103 - 107 °C (Expl.) |
| Boiling point | 228.8±20.0 °C 760 mmHg (Calc.)* |
| Flash point | 92.2±21.8 °C (Calc.)* |
| Index of refraction | 1.612 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
5-Bromonicotinonitrile is an organic compound that belongs to the class of halogenated pyridine derivatives. It consists of a pyridine ring, a nitrile group (–C≡N) at the 2-position, and a bromine atom at the 5-position on the pyridine ring. The molecular structure of 5-bromonicotinonitrile combines the characteristics of both a nitrile group and a halogen-substituted aromatic ring, making it an important intermediate for various chemical syntheses. This compound is particularly useful in the synthesis of other organic molecules, including bioactive compounds. The nitrile group, a key functional group, is often employed in reactions that form carbon-carbon bonds and can be easily converted into other functional groups, such as amides, acids, or alcohols. The bromine atom, positioned at the 5-position of the pyridine ring, makes the compound reactive in several important organic reactions, particularly in nucleophilic substitution reactions and cross-coupling reactions. This makes 5-bromonicotinonitrile a useful intermediate in the synthesis of more complex organic molecules, including pharmaceuticals and agrochemicals. 5-Bromonicotinonitrile has been explored in the development of compounds with potential biological activity. Pyridine and nitrile-containing compounds, in general, exhibit a wide range of biological effects, such as antimicrobial, anticancer, and anti-inflammatory activities. The presence of the bromine atom further enhances the reactivity and versatility of the compound, allowing for the introduction of additional functional groups or the creation of novel molecular frameworks with potential therapeutic properties. Additionally, 5-bromonicotinonitrile is used in materials science and as a building block for the synthesis of specialized ligands in coordination chemistry. It can also be employed in the preparation of conducting polymers and other materials with electronic properties. In summary, 5-bromonicotinonitrile is a versatile halogenated pyridine derivative with a nitrile functional group that makes it useful in a variety of organic syntheses. It is an important intermediate in the preparation of pharmaceuticals, agrochemicals, and materials, and its bromine atom provides reactivity that enables its use in a range of chemical reactions. The compound's potential biological activity and its role in synthetic chemistry highlight its importance in both academic and industrial research. References 2010. Preparation of functionalized organoaluminiums by direct insertion of aluminium to unsaturated halides. Nature Chemistry, 2(4). DOI: 10.1038/nchem.590 2017. Synthetic approaches toward stilbenes and their related structures. Molecular Diversity, 21(4). DOI: 10.1007/s11030-017-9736-9 2013. Convenient Synthesis of Benzoxazolone Derivatives by Cross-Coupling of Benzoxazolone Boronates with Aryl Halides. Synthesis, 45(22). DOI: 10.1055/s-0033-1339799 |
| Market Analysis Reports |