Online Database of Chemicals from Around the World
| Shanghai Medpep Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.medpep.com | |||
 | +86 (21) 6556-6949 | |||
 | +86 (21) 6556-9401 | |||
 | medpep@gmail.com xueminguo@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jiangxi Kingnord Industrial Limited | China | |||
|---|---|---|---|---|
 | www.kingnordchem.com | |||
 | +86 (791) 8649-3591 | |||
 | +86 (791) 8649-3642 | |||
 | jeff@kingnord.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Enki Biopharmaceuticals (Shanghai) Limited | China | |||
|---|---|---|---|---|
 | www.enkibiopharma.com | |||
 | +86 (21) 5768-0965 +86 13916707528 | |||
 | +86 (21) 5768-0922 | |||
 | info@enkibiopharma.com | |||
 | QQ Chat | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Shanghai GL Synthesis Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.glsyn.com | |||
 | +86 (21) 5409-1896 | |||
 | +86 (21) 5409-1896 / 6318-6607 | |||
 | charlie@glsyn.com | |||
| Chemical manufacturer since 2000 | ||||
| Endeavour Speciality Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.endeavourchem.co.uk | |||
 | +44 (1327) 310-079 | |||
 | +44 (1327) 310-701 | |||
 | enquiries@endeavourchem.co.uk | |||
| Chemical manufacturer | ||||
| GLSynthesis Inc. | USA | |||
|---|---|---|---|---|
 | www.glsynthesis.com | |||
 | +1 (508) 754-6700 | |||
 | +1 (508) 754-7075 | |||
 | support@glsynthesis.com | |||
| Chemical manufacturer | ||||
| Watanabe Chemical Ind., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.watanabechem.co.jp | |||
 | +81 (82) 231-0540 | |||
 | +81 (82) 231-1451 | |||
 | inquiry@watanabechem.co.jp | |||
| Chemical manufacturer | ||||
| Dalian Honkai Chemical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.honkai.com | |||
 | +86 (411) 8479-1969 | |||
 | +86 (411) 8482-0706 | |||
 | sales@honkai.com | |||
| Chemical manufacturer since 2005 | ||||
| IRIS Biotech GmbH | Germany | |||
|---|---|---|---|---|
 | www.iris-biotech.de | |||
 | +49 (9231) 961-973 | |||
 | +49 (9231) 961-999 | |||
 | info@iris-biotech.de | |||
| Chemical manufacturer since 1788 | ||||
| Classification | Chemical reagent >> Organic reagent >> Ester >> Butyl ester compound |
|---|---|
| Name | tert-Butyl N-hydroxycarbamate |
| Synonyms | N-Boc-hydroxylamine |
| Molecular Structure | 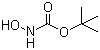 |
| Molecular Formula | C5H11NO3 |
| Molecular Weight | 133.15 |
| CAS Registry Number | 36016-38-3 |
| EC Number | 252-836-2 |
| SMILES | CC(C)(C)OC(=O)NO |
| Melting point | 53-57 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
tert-Butyl N-hydroxycarbamate, also known as Boc-NHOH, was first synthesized during investigations into protecting amine groups in organic synthesis. Chemists sought to develop stable protecting groups that could be easily removed under mild conditions to allow for selective functionalization of amines. The synthesis involved the reaction of tert-butylamine with chloroformates and hydroxylamine to form the N-hydroxycarbamate derivative. This discovery represented a significant advancement in organic chemistry, providing a versatile tool for selectively protecting amino groups in complex molecules. tert-Butyl N-hydroxycarbamate is used as a protecting group for amines in organic synthesis, preventing unwanted reactions with other reagents while allowing desired transformations elsewhere in the molecule. This strategy is crucial in multi-step synthesis to control regioselectivity and achieve complex molecular architectures. After desired transformations, tert-Butyl N-hydroxycarbamate can be selectively removed under mild conditions to regenerate the free amine group, enabling the recovery of the original functionality without affecting other sensitive groups present. tert-Butyl N-hydroxycarbamate is used in solid-phase peptide synthesis to protect the N-terminus of growing peptide chains, enabling efficient and high-yield synthesis of peptides and peptidomimetics for pharmaceutical research. It serves as part of orthogonal protecting group strategies in peptide synthesis, allowing precise control over peptide assembly for the synthesis of complex peptide structures with specific functionalities. tert-Butyl N-hydroxycarbamate plays a crucial role in synthesizing pharmaceutical intermediates and active pharmaceutical ingredients (APIs), contributing to the development of new drug candidates with improved properties. It can be incorporated into prodrugs to improve drug stability, bioavailability, and tissue targeting, making it valuable in drug delivery systems. In materials science, tert-Butyl N-hydroxycarbamate is used to modify surfaces and polymers, enhancing properties such as adhesion, corrosion resistance, and biocompatibility in coatings, adhesives, and biomedical devices. It is employed in polymer chemistry for controlled modification of polymer chains, allowing tailored properties such as solubility, thermal stability, and mechanical strength, expanding applications in drug delivery and nanotechnology. References 2019. Hydroxylamine Derivatives in Synthesis. Organic & Biomolecular Chemistry, 17(28). DOI: 10.1039/C9OB00987A |
| Market Analysis Reports |