Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Watec Laboratories, Inc. | China | |||
|---|---|---|---|---|
 | www.wateclaboratories.com | |||
 | +86 (519) 8692-1516 +86 18602586511 | |||
 | +86 (519) 8526-7382 | |||
 | info@wateclaboratories.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives >> Carboxylic esters and their derivatives |
|---|---|
| Name | tert-Butyl ((1S,2R)-2-aminocyclohexyl)carbamate |
| Synonyms | tert-Butyl N-((2R,1S)-2-aminocyclohexyl)carbamate; ((1S,2R)-2-Aminocyclohexyl)carbamic acid tert-butyl ester |
| Molecular Structure | 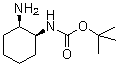 |
| Molecular Formula | C11H22N2O2 |
| Molecular Weight | 214.30 |
| CAS Registry Number | 365996-30-1 |
| EC Number | 690-960-2 |
| SMILES | CC(C)(C)OC(=O)N[C@H]1CCCC[C@H]1N |
| Density | 1.0$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 322.1$+/-$31.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 148.6$+/-$24.8 $degree$C (Calc.)* |
| Index of refraction | 1.488 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H318 Details | ||||||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P354+P338-P317-P321-P332+P317-P362+P364 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
tert-Butyl ((1S,2R)-2-aminocyclohexyl)carbamate is a chiral carbamate-protected cyclohexylamine derivative with the molecular formula C11H22N2O2. It consists of a cyclohexane ring bearing an amino substituent and a carbamate protecting group, specifically a tert-butoxycarbonyl (Boc) group attached through a carbamate linkage. The compound contains two stereogenic centers at the 1 and 2 positions of the cyclohexane ring, defined by the (1S,2R) configuration. Structurally, the molecule is derived from 2-aminocyclohexanol or related cyclohexylamine precursors in which the amino group is protected as a Boc-carbamate. The tert-butyl carbamate group has the general structure –NH–C(=O)–O–C(CH3)3, where the carbonyl carbon is linked to the nitrogen of the cyclohexylamine and the oxygen is bonded to a tert-butyl group. This protecting group is widely used in organic synthesis to temporarily mask amine functionality. The cyclohexane ring adopts conformations typical of six-membered saturated rings, most commonly the chair form. The stereochemistry at positions 1 and 2 strongly influences the spatial orientation of substituents, which in turn affects reactivity and interactions in synthetic applications. The (1S,2R) configuration defines a specific three-dimensional arrangement that can be important in asymmetric synthesis or chiral intermediate preparation. The Boc (tert-butoxycarbonyl) protecting group is stable under neutral and basic conditions but can be removed under acidic conditions, typically using strong acids such as trifluoroacetic acid or hydrochloric acid. This acid-labile property makes it one of the most widely used amine protecting groups in peptide synthesis and medicinal chemistry. The amino group in the molecule is a primary amine prior to protection and can participate in nucleophilic reactions, condensation reactions with carbonyl compounds, and coordination with metal ions. However, in the carbamate-protected form, its reactivity is suppressed, allowing selective transformations elsewhere in a synthetic sequence. tert-Butyl ((1S,2R)-2-aminocyclohexyl)carbamate is typically used as an intermediate in organic synthesis, particularly in the preparation of more complex nitrogen-containing molecules. Chiral cyclohexylamine derivatives are valuable building blocks in pharmaceutical and asymmetric synthesis due to their rigid ring structure and defined stereochemistry. The synthesis of such compounds generally involves selective protection of cyclohexylamine derivatives using di-tert-butyl dicarbonate (Boc2O) under mild basic conditions. The stereochemical integrity of the cyclohexane framework is preserved during protection, allowing the compound to serve as a chiral intermediate. From a physicochemical perspective, the molecule exhibits moderate polarity due to the carbamate functional group, while the cyclohexane ring and tert-butyl group contribute hydrophobic character. The compound can participate in hydrogen bonding through the carbamate NH and carbonyl oxygen atoms. The presence of both stereocenters and a protected amine makes this compound useful in stereochemically controlled synthesis. It can serve as a precursor for further functionalization, including deprotection to regenerate the free amine or substitution reactions on the cyclohexane framework. Historically, Boc-protected amines have become standard tools in synthetic organic chemistry due to their stability and ease of installation and removal. Compounds such as tert-butyl ((1S,2R)-2-aminocyclohexyl)carbamate represent common intermediates in the synthesis of chiral amines, ligands, and pharmaceutical building blocks. Overall, tert-butyl ((1S,2R)-2-aminocyclohexyl)carbamate is a stereochemically defined Boc-protected cyclohexylamine derivative. Its combination of rigid chiral framework and acid-labile amine protection makes it a useful intermediate in asymmetric synthesis and organic chemistry applications. References 2025. Preparation method of cis- (1S, 2R) -N-tert-butoxycarbonyl-1, 2-cyclohexanediamine. CN-116836032-B. URL: https://patents.google.com/patent/CN116836032B |
| Market Analysis Reports |