Lithium bis(trimethylsilyl)amide (LiHMDS) is a widely used organolithium reagent that plays a significant role in organic synthesis, particularly in the formation of carbon-carbon and carbon-heteroatom bonds. This chemical compound consists of a lithium cation and the bis(trimethylsilyl)amide anion, a highly reactive species. LiHMDS is especially notable for its strong basicity, which allows it to deprotonate weak acids, making it an essential tool in a variety of synthetic processes.
The discovery of lithium bis(trimethylsilyl)amide is attributed to advancements in the synthesis and application of organolithium reagents, which were first developed in the early 20th century. Organolithium compounds, particularly lithium amides, have long been used in synthetic chemistry due to their ability to form nucleophilic intermediates. LiHMDS was introduced as a more stable and less reactive alternative to other lithium amides, such as lithium diisopropylamide (LDA), providing improved selectivity and ease of handling.
LiHMDS is synthesized by reacting lithium metal with bis(trimethylsilyl)amine in anhydrous solvents, typically tetrahydrofuran (THF) or diethyl ether, under an inert atmosphere to prevent decomposition. The resulting reagent is highly soluble in these nonpolar solvents, making it ideal for use in a wide range of reactions.
The primary application of LiHMDS lies in its use as a strong base in organic reactions, particularly in the deprotonation of carbon-hydrogen bonds. Its basicity allows it to selectively deprotonate compounds that are otherwise difficult to react with. This characteristic is particularly valuable in the synthesis of enolates, which are key intermediates in many organic transformations, including aldol condensations and Michael additions. LiHMDS is also used to generate a variety of reactive intermediates, such as alkyl and aryl lithium compounds, which can undergo further transformations to form complex molecules.
One of the major applications of LiHMDS is in the synthesis of carbon-carbon bonds, specifically in the formation of enolates, which can be used in a variety of reactions, including the alkylation of carbonyl compounds, ester enolization, and nucleophilic additions. This is particularly important in the synthesis of pharmaceuticals and agrochemicals, where the ability to form complex carbon-carbon bonds with high selectivity is a key aspect of drug development.
LiHMDS is also employed in the preparation of other important intermediates, such as organozinc and organoboron compounds, which are used in a variety of cross-coupling reactions, including Suzuki, Negishi, and Kumada couplings. These reactions are widely utilized in the synthesis of a range of biologically active molecules, including natural products and pharmaceutical candidates.
In addition to its role in carbon-carbon bond formation, LiHMDS is used in the synthesis of compounds containing heteroatoms, such as nitrogen, oxygen, and sulfur. For example, it is used in the synthesis of amides, ketones, and lactones, which are common structural motifs in pharmaceuticals and other biologically active compounds.
Lithium bis(trimethylsilyl)amide is also useful in the synthesis of functionalized silanes and siloxanes, which have applications in materials science, particularly in the production of specialty polymers and coatings. Its ability to activate various substrates makes it a valuable reagent in the creation of novel materials, such as siloxane-based polymers, which are used in electronics, coatings, and adhesives.
The stability and ease of use of LiHMDS make it a valuable reagent in both academic and industrial research. However, its highly reactive nature means it must be handled with care, particularly when working with moisture-sensitive reactions. Proper storage in inert, dry conditions and the use of anhydrous solvents are essential to prevent degradation and side reactions.
References
2024. Nucleophilic trifluoromethylation with CF3H/LiHMDS: probing the nucleophilic reactivity of LiCF3 species. Science China Chemistry, 67(9).
DOI: 10.1007/s11426-024-2289-7
1975. Synthesis of N-trimethylsilyl-substituted organoaminosilanes and cyclodisilazanes. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 24(11).
DOI: 10.1007/bf00921677
|












































































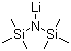


 GHS02;GHS05;GHS07 Danger Details
GHS02;GHS05;GHS07 Danger Details