Lithium bis(trifluoromethanesulphonyl)imide (LiTFSI) is a highly important lithium salt that has garnered attention for its role in the development of advanced batteries and electrochemical systems. This compound, known for its chemical stability and high ionic conductivity, was first synthesized and introduced in the late 20th century as part of the ongoing search for more efficient electrolytes in lithium-based energy storage systems. LiTFSI has since become a key component in the development of lithium-ion batteries, supercapacitors, and other electrochemical devices, where its unique properties enable enhanced performance and stability.
The discovery of LiTFSI can be traced back to the exploration of fluorinated anions for use in battery electrolytes. Researchers were seeking alternatives to lithium salts like lithium hexafluorophosphate (LiPF6), which, while widely used, had limitations in terms of thermal stability and moisture sensitivity. In the 1980s and 1990s, scientists synthesized LiTFSI, characterized by its large, non-coordinating TFSI anion. This anion features two trifluoromethanesulfonyl groups attached to nitrogen, forming a structure that enhances solubility and stability in a variety of organic solvents while minimizing ion pairing. The result is a lithium salt that exhibits excellent electrochemical performance under a wide range of conditions.
One of the key applications of LiTFSI is in lithium-ion batteries, where it serves as an electrolyte additive or as the primary lithium salt in the electrolyte solution. Lithium-ion batteries have become the dominant energy storage technology in consumer electronics, electric vehicles, and renewable energy storage due to their high energy density, long cycle life, and low self-discharge rate. LiTFSI is preferred in these systems because it offers superior thermal and chemical stability compared to traditional salts like LiPF6. This stability is crucial for improving the safety and longevity of batteries, particularly in high-temperature environments or applications where safety is a top priority.
LiTFSI is also known for its high ionic conductivity, which plays a critical role in improving the efficiency of lithium-ion batteries. In an electrochemical cell, the movement of lithium ions between the anode and cathode is essential for charge and discharge cycles. LiTFSI facilitates this process by providing a highly conductive medium that allows lithium ions to move more freely, thus enhancing the overall performance of the battery. Its compatibility with a wide range of solvents, including both organic carbonates and ionic liquids, further broadens its applicability in different battery designs, including high-voltage and solid-state batteries.
Beyond lithium-ion batteries, LiTFSI has found applications in supercapacitors and other energy storage devices. Supercapacitors, which offer high power density and fast charge-discharge cycles, benefit from the use of LiTFSI due to its excellent conductivity and thermal stability. The salt’s ability to operate effectively in extreme conditions makes it ideal for applications where rapid energy delivery is required, such as in automotive systems and industrial machinery. Moreover, its use in combination with ionic liquids has opened up new possibilities for developing safer and more efficient supercapacitor technologies.
LiTFSI also plays an important role in solid polymer electrolytes, which are being explored as safer alternatives to liquid electrolytes in next-generation battery technologies. In solid-state batteries, LiTFSI is often used to improve the ionic conductivity of polymer matrices, enabling the development of more robust and less flammable batteries. Solid-state batteries are of particular interest due to their potential to offer higher energy densities and improved safety compared to conventional lithium-ion batteries, and LiTFSI is a critical component in making these advancements possible.
In addition to its applications in energy storage, LiTFSI has been studied for use in electrochemical devices like fuel cells, where it can serve as a supporting electrolyte due to its chemical inertness and conductivity. Its high electrochemical stability window makes it suitable for use in harsh environments, allowing for the development of more durable and long-lasting fuel cell systems.
In conclusion, the discovery of lithium bis(trifluoromethanesulphonyl)imide has had a profound impact on the field of energy storage and electrochemistry. Its unique properties, including high ionic conductivity, thermal stability, and compatibility with a wide range of solvents, have made it a crucial component in lithium-ion batteries, supercapacitors, and solid-state batteries. As the demand for more efficient and safer energy storage technologies continues to grow, LiTFSI will remain an important material in the advancement of next-generation electrochemical systems.
References
2024. Extraction of U(VI), Th(IV), and Lanthanides(III) from Nitric Acid Solutions with Mixtures of Diphenyl-N,N-dioctylcarbamoylmethylphosphine Oxide and Lithium Bis[(trifluoromethyl)sulfonyl]imide. Radiochemistry, 66(5).
DOI: 10.1134/s1066362224050060
2019. Stabilized iron fluoride cathodes. Nature Materials, 18(12).
DOI: 10.1038/s41563-019-0540-z
2016. A novel quasi-solid state electrolyte with highly effective polysulfide diffusion inhibition for lithium-sulfur batteries. Scientific Reports, 6(1).
DOI: 10.1038/srep25484
|






























































































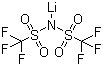


 GHS06;GHS05;GHS08 Danger Details
GHS06;GHS05;GHS08 Danger Details