Online Database of Chemicals from Around the World
| Hangzhou Greenda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.greendachem.com | |||
 | +86 (571) 8777-4478 | |||
 | +86 (571) 8777-4347 | |||
 | greenda@greendachem.com gongsh1018@126.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Futoh Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.futoh.co.jp | |||
 | +81 (52) 521-8171 | |||
 | +81 (52) 521-8793 | |||
 | tokoro@futoh.co.jp | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Hangzhou Bio-and Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.baokaichem.com | |||
 | +86 (571) 8502-9599 | |||
 | +86 (571) 8502-9600 | |||
 | sales@baokaichem.com | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Survival Technologies Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.survivaltechnologies.in | |||
 | +91 (22) 4212-8221 | |||
 | +91 (22) 4212-8226 | |||
 | info@survivaltechnologies.in | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Jinan Wanxingda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wxdchem.com | |||
 | +86 (531) 8806-4106 +86 18253116016 | |||
 | +86 (531) 8806-4109 | |||
 | bruce@wxdchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Reddy N Reddy Pharmaceuticals | India | |||
|---|---|---|---|---|
 | www.reddynreddypharma.com | |||
 | +91 (40) 2307-5686 +91 98480-96759 +91 98481-96759 | |||
 | info@reddynreddypharma.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Jiangxi Time Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.groupchem.com | |||
 | +86 (794) 718-3888 718-3887 526-9970 +86 13867635890 | |||
 | +86 (794) 526-9668 / 526-9166 | |||
 | sales@groupchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Haimen RuiYi Medical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruiyitech.cn | |||
 | +86 (21) 5471-1706 | |||
 | +86 (21) 5471-1707 | |||
 | sales@ruiyitech.com zyj3625@msn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Jinan Great Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemgreat.com | |||
 | +86 (531) 5866-8191 | |||
 | +86 (531) 8632-7119 | |||
 | sale001@chemgreat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Qinba Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shangfluoro.com | |||
 | +86 (21) 6517-0832 +86 17301729617 | |||
 | +86 (21) 6517-0832 | |||
 | shangfluoro@hotmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Tianwei Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianweipharm.com | |||
 | +86 (571) 8516-6702 8516-6621 | |||
 | sales@tianweipharm.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Xiamen AmoyChem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amoychem.com | |||
 | +86 (592) 605-1114 +86 18959220845 | |||
 | +86 (592) 605-1114 | |||
 | sales@amoychem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Shandong Zhishang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhishangchemical.com | |||
 | +86 17653113209 13065062079 | |||
 | sales001@sdzschem.com sales002@sdzschem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Xinlian Rui Biomedical Technology (shandong) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xlrchem.com | |||
 | +86 15153173989 | |||
 | 3865799121@qq.com | |||
 | QQ Chat | |||
 | WeChat: 15153173989 | |||
| Chemical manufacturer since 2024 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Sinbond Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinbondpharm.com | |||
 | +86 (531) 8703-8285 +86 13583181986 +44 (208) 242-5518 | |||
 | +86 (531) 8703-8285 | |||
 | cici1124@gmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2014 | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Oakwood Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.oakwoodchemical.com | |||
 | +1 (800) 467-3386 | |||
 | k_weaver@oakwoodchemical.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical pesticide >> Insecticide intermediate |
|---|---|
| Name | Trifluoroacetic anhydride |
| Synonyms | 2,2,2-Trifluoroacetic anhydride; TFAA |
| Molecular Structure | 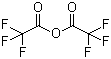 |
| Molecular Formula | C4F6O3 |
| Molecular Weight | 210.03 |
| CAS Registry Number | 407-25-0 |
| EC Number | 206-982-9 |
| SMILES | C(=O)(C(F)(F)F)OC(=O)C(F)(F)F |
| Density | 1.6±0.1 g/cm3 Calc.*, 1.511 g/mL (Expl.) |
|---|---|
| Melting point | -65 °C (Expl.) |
| Boiling point | 39.5 °C 760 mmHg (Calc.)*, 39.5 - 40 °C (Expl.) |
| Flash point | -26.1±20.8 °C (Calc.)* |
| Solubility | water hydrolysis (Expl.) |
| Index of refraction | 1.291 (Calc.)*, 1.269 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS07;GHS08 Danger Details GHS05;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H318-H332-H361d-H412 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P264-P264+P265-P271-P273-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P318-P321-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1760; UN 3265 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Trifluoroacetic anhydride is a highly reactive organofluorine compound with the molecular formula (CF3CO)2O. It is the anhydride of trifluoroacetic acid and was first developed in the mid-20th century as chemists explored the synthesis and applications of fluorinated reagents. The presence of two trifluoromethyl groups strongly increases the electrophilicity of the carbonyl carbons, making the anhydride significantly more reactive than the parent acid. Trifluoroacetic anhydride is typically a colorless liquid that is highly moisture-sensitive and must be handled under anhydrous conditions to prevent hydrolysis. The primary application of trifluoroacetic anhydride is as a reagent for introducing the trifluoroacetyl group into nucleophilic substrates such as amines, alcohols, and thiols. This reaction forms trifluoroacetamides, esters, and thioesters, which are important intermediates in organic synthesis. The trifluoromethyl group imparts strong electron-withdrawing effects, stabilizing reaction intermediates and altering chemical reactivity. Its ability to selectively introduce the trifluoroacetyl group has made it a valuable tool in the preparation of fluorinated building blocks used in pharmaceuticals, agrochemicals, and specialty chemicals. In pharmaceutical chemistry, trifluoroacetic anhydride is widely used to synthesize trifluoromethylated derivatives of bioactive molecules. Incorporation of the trifluoroacetyl group can enhance metabolic stability, lipophilicity, and pharmacokinetic properties of drug candidates. It has been employed in the synthesis of enzyme inhibitors, antiviral agents, and central nervous system drugs, where the trifluoromethyl moiety improves biological activity and chemical robustness. Its high reactivity enables efficient functionalization under mild conditions, which is important for sensitive or complex molecules. In agrochemical research, trifluoroacetic anhydride is used to prepare trifluoromethylated herbicides, fungicides, and insecticides. The trifluoromethyl group improves chemical and thermal stability, environmental persistence, and lipophilicity, enhancing the efficacy of the final products. The anhydride allows chemists to introduce this group efficiently into precursor molecules, facilitating the development of new biologically active compounds. Trifluoroacetic anhydride also finds application in materials science and polymer chemistry. Fluorinated acyl groups introduced using the anhydride increase the hydrophobicity, chemical resistance, and thermal stability of polymers and specialty coatings. Its use in selective functionalization reactions provides a way to modify material properties without compromising other sensitive substituents. Analytical chemistry benefits from trifluoroacetic anhydride as well. It is sometimes used as a derivatization reagent for improving the volatility and detectability of polar compounds in gas chromatography and mass spectrometry. The trifluoroacetyl derivatives formed are more volatile and thermally stable, allowing for better separation and detection. The discovery and continued use of trifluoroacetic anhydride underscore its importance as a reactive fluorinated reagent. Its combination of strong electrophilicity, stability, and versatility has made it a cornerstone in organic synthesis, supporting advances in pharmaceuticals, agrochemicals, materials science, and analytical chemistry. The compound exemplifies the value of fluorinated anhydrides in modern chemical research and industrial applications. References 2013. Synthesis of 2-Nitroglycals from Glycals Using the Tetrabutylammonium Nitrate-Trifluoroacetic Anhydride-Triethylamine Reagent System and Base-Catalyzed Ferrier Rearrangement of Acetylated 2-Nitroglycals. The Journal of Organic Chemistry, 78(16). DOI: 10.1021/jo401165y 2012. Production of identical retention times and mass spectra for δ9-tetrahydrocannabinol and cannabidiol following derivatization with trifluoroacetic anhydride with 1,1,1,3,3,3-hexafluoroisopropanol. Journal of Analytical Toxicology, 36(1). DOI: 10.1093/jat/bkr017 1984. Electron-Capture Capillary Gas Chromatographic Determination of Phenylpropanolamine in Human Plasma Following Derivatization with Trifluoroacetic Anhydride. Journal of Pharmaceutical Sciences, 73(9). DOI: 10.1002/jps.2600730936 |
| Market Analysis Reports |