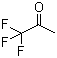
1,1,1-Trifluoroacetone (C3HF3CO) is an organic compound containing both a trifluoromethyl group and an acetone structure. It is a colorless liquid with a sharp, pungent odor and is highly reactive due to the presence of the trifluoromethyl group. The trifluoromethyl group, characterized by its electron-withdrawing properties, imparts unique chemical reactivity to the compound, making it useful in various industrial and synthetic applications.
The discovery of 1,1,1-trifluoroacetone dates back to the mid-20th century, with its synthesis being a topic of interest in the study of fluorinated compounds. The compound is typically synthesized through the halogenation of acetone, where fluorine is introduced into the molecule, specifically replacing one of the hydrogen atoms with a trifluoromethyl group. This reaction results in the formation of 1,1,1-trifluoroacetone, which can then be isolated and purified for various uses.
One of the primary applications of 1,1,1-trifluoroacetone is in the synthesis of fluorinated chemicals. The trifluoromethyl group present in the compound is a valuable building block in organic synthesis, as it enhances the chemical stability, lipophilicity, and reactivity of molecules. This makes it useful in the development of pharmaceuticals, agrochemicals, and specialty chemicals. The introduction of a trifluoromethyl group into organic molecules often results in improved bioactivity and altered chemical properties, which can enhance the performance of the final products.
In the pharmaceutical industry, 1,1,1-trifluoroacetone is used as an intermediate in the synthesis of fluorinated drug candidates. Fluorinated drugs are of particular interest due to their enhanced metabolic stability, improved binding affinity to target proteins, and the potential for better pharmacokinetic properties. The trifluoromethyl group is often incorporated into drugs to modify their interactions with biological systems, making them more effective in treating a variety of conditions, including cancers and cardiovascular diseases.
1,1,1-Trifluoroacetone is also employed in the synthesis of agrochemicals, particularly in the development of herbicides and insecticides. The presence of the trifluoromethyl group in agrochemical molecules can improve their potency, stability, and resistance to degradation. This results in more effective and longer-lasting pest control, which is beneficial in agricultural practices.
Additionally, 1,1,1-trifluoroacetone is utilized in materials science, particularly in the development of high-performance materials that require enhanced chemical resistance, stability, and durability. The incorporation of the trifluoromethyl group into polymers and coatings imparts desirable properties such as hydrophobicity, resistance to solvents, and improved weathering resistance. These materials find applications in industries such as electronics, automotive, and aerospace, where high-performance materials are essential.
Due to the reactivity and potential hazards associated with the trifluoromethyl group, 1,1,1-trifluoroacetone must be handled with care. It is important to consider the environmental and safety implications of its use, as fluorinated compounds can persist in the environment and may be toxic. Regulatory measures for the safe handling, use, and disposal of 1,1,1-trifluoroacetone are important to minimize its ecological impact.
In conclusion, 1,1,1-trifluoroacetone is a versatile fluorinated compound with applications in organic synthesis, pharmaceuticals, agrochemicals, and materials science. Its trifluoromethyl group provides unique chemical properties that make it a valuable intermediate for the development of various industrial products. However, due to its reactivity and potential environmental impact, proper handling and regulatory oversight are essential for its safe use.
References
2022. Enantiopure Β3-Trifluoromethyl-Β3-homoalanine Derivatives: Coupling with Boc-Protected Amino Acids and Conformational Studies of Peptides in Solid State. Synthesis, 54(12).
DOI: 10.1055/s-0041-1737396
2011. Straightforward synthesis of 1-alkyl-2-(trifluoromethyl)aziridines starting from 1,1,1-trifluoroacetone. Org. Biomol. Chem., 9(20).
DOI: 10.1039/c1ob05813d
|


































































 GHS02;GHS05;GHS07 Danger Details
GHS02;GHS05;GHS07 Danger Details