Online Database of Chemicals from Around the World
| Epochem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.epochem.com | |||
 | +86 (21) 6760-1595 6760-1597 | |||
 | +86 (21) 6760-1605 | |||
 | seth_wang@epochem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2005 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Zhejiang Chempharm Industry&trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chempharm.cn | |||
 | +86 (571) 8770-1568 | |||
 | export@chempharm.cn | |||
| Chemical distributor since 1996 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Creative Peptides | USA | |||
|---|---|---|---|---|
 | www.creative-peptides.com | |||
 | +1 (631) 624-4882 | |||
 | +1 (631) 614-7828 | |||
 | info@creative-peptides.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Evershine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.evershinechem.net | |||
 | +86 (571) 5812-0272 5812-0273 | |||
 | +86 (571) 5812-0272 | |||
 | sales@evershinechem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Cherry Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.cherrypharmatech.com | |||
 | +86 (571) 8163-6070 +86 18042403330 | |||
 | +86 (571) 8163-6070 | |||
 | info@cherrypharmatech.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Watanabe Chemical Ind., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.watanabechem.co.jp | |||
 | +81 (82) 231-0540 | |||
 | +81 (82) 231-1451 | |||
 | inquiry@watanabechem.co.jp | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Proline derivatives |
|---|---|
| Name | (S)-2-Methylproline |
| Synonyms | (S)-2-Methylpyrrolidine-2-carboxylic acid |
| Molecular Structure | 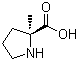 |
| Molecular Formula | C6H11NO2 |
| Molecular Weight | 129.16 |
| Protein Sequence | X |
| CAS Registry Number | 42856-71-3 |
| EC Number | 610-068-9 |
| SMILES | C[C@]1(CCCN1)C(=O)O |
| Melting point | 310 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
(S)-2-Methylproline is an amino acid derivative that is notable for its chiral center and unique structural features. As a member of the proline family, it has attracted attention from various scientific fields due to its unique properties and wide range of applications. (S)-2-Methylproline was first synthesized in the late 20th century when researchers were exploring proline analogs with altered side chains. The goal was to modify the structure of proline to enhance its biological activity or improve its utility in chemical synthesis. The introduction of a methyl group at the 2-position of the proline ring generates (S)-2-methylproline, which exhibits interesting chemical and biological properties. Its synthesis has provided valuable insights into the effects of substituents on the behavior and function of the proline ring. In medicinal chemistry, (S)-2-methylproline is used as a chiral building block for drug development. Its unique structure can influence the pharmacological properties of drugs, making it a useful component for the design of new drugs. Researchers have exploited its chirality to create compounds with specific biological activities, thereby improving the efficacy and selectivity of therapeutic agents. (S)-2-Methylproline is a valuable intermediate in organic synthesis. Its presence in synthetic pathways can facilitate the construction of complex molecules. The compound is used to develop new materials, fine chemicals, and other organic compounds, taking advantage of its structural features to achieve desired reactivity and properties. In biochemical research, (S)-2-methylproline is used to study the effects of proline derivatives on protein folding and function. Its effects on protein stability and conformation can provide insights into protein chemistry and aid in the design of proteins with tailored properties for a variety of applications. The compound is also explored in catalytic processes. Its chiral centers make it useful as a chiral ligand or catalyst in asymmetric syntheses. (S)-2-methylproline can aid in the development of chiral catalysts that produce enantiomerically pure compounds, which are key to many chemical and pharmaceutical processes. References 1976. Stereochemistry of the enzymic synthesis of the tert.-leucine side chain. Helvetica Chimica Acta, 59(5). DOI: 10.1002/hlca.19760590523 2005. Enantioselective addition of diethylzinc to aldehydes catalyzed by (S)-2-(arylhydroxymethyl)-1-methylpyrrolidine. Tetrahedron: Asymmetry, 16(3). DOI: 10.1016/j.tetasy.2004.12.017 2024. Rett syndrome. Nature Reviews Disease Primers, 10(1). DOI: 10.1038/s41572-024-00568-0 |
| Market Analysis Reports |