Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Survival Technologies Pvt Ltd | India | |||
|---|---|---|---|---|
 | www.survivaltechnologies.in | |||
 | +91 (22) 4212-8221 | |||
 | +91 (22) 4212-8226 | |||
 | info@survivaltechnologies.in | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Sifluorotm Co,. Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoro.com | |||
 | +86 (0531) 8590-6692 | |||
 | +86 (0531) 8590-6692 | |||
 | inquiry@fluoro.com | |||
 | WeChat: 15288857388 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2025 | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| Rieke Metals, Inc. | USA | |||
|---|---|---|---|---|
 | www.riekemetals.com | |||
 | +1 (402) 434-2775 | |||
 | +1 (402) 434-2777 | |||
 | sales@riekemetals.com | |||
| Chemical manufacturer | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | Trifluoroacetophenone |
| Synonyms | 2,2,2-Trifluoroacetophenone |
| Molecular Structure | 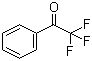 |
| Molecular Formula | C8H5F3O |
| Molecular Weight | 174.12 |
| CAS Registry Number | 434-45-7 |
| EC Number | 207-103-1 |
| SMILES | C1=CC=C(C=C1)C(=O)C(F)(F)F |
| Density | 1.3±0.1 g/cm3, Calc.*, 1.278 g/mL (Expl.), 1.458 (Expl.) |
|---|---|
| Melting point | 51-40 °C (Expl.) |
| Index of Refraction | 1.448, Calc.* |
| Boiling Point | 165.5 °C (760 mmHg), Calc.*, 165-166 °C (Expl.) |
| Flash Point | 41.7 °C, Calc.*, 41 °C (Expl.) |
| Water solubility | PRACTICALLY INSOLUBLE |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS02;GHS07 Warning Details GHS02;GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P271-P280-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 1224 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Trifluoroacetophenone (C8H7F3O) is an organic compound that consists of a phenyl group attached to a trifluoroacetyl group, making it a member of the acetophenone family. This compound is known for its distinctive structure, which includes three fluorine atoms bonded to the carbon adjacent to the carbonyl group. It is a colorless to light yellow liquid with a pleasant, sweet odor and is mainly used in the field of organic synthesis. Trifluoroacetophenone was first synthesized and characterized in the mid-20th century, with interest primarily in its applications as an intermediate for chemical synthesis. The process of synthesizing trifluoroacetophenone typically involves the reaction of acetophenone with a suitable source of trifluoroacetic acid, such as trifluoroacetic anhydride or trifluoroacetyl chloride, under anhydrous conditions. This reaction introduces the trifluoromethyl group at the para-position of the aromatic ring relative to the carbonyl group. The unique properties of trifluoroacetophenone are largely attributed to the presence of the trifluoromethyl group (-CF3). The electronegativity of fluorine atoms, particularly in the trifluoromethyl group, imparts special chemical reactivity to the molecule. This modification can enhance the compound's ability to participate in various chemical reactions, particularly those involving nucleophilic attack, electrophilic substitution, and coordination with metal centers. Additionally, the trifluoromethyl group increases the compound's stability and can affect its polarity and solubility, which are beneficial in some applications. One of the primary applications of trifluoroacetophenone is in the synthesis of pharmaceuticals. The trifluoromethyl group can significantly impact the biological activity and pharmacological properties of drugs, including their metabolic stability, lipophilicity, and interaction with specific receptors. Trifluoroacetophenone is used as an intermediate in the preparation of a range of bioactive molecules. The compound's ability to influence drug design, particularly in enhancing the potency or selectivity of therapeutic agents, makes it valuable in medicinal chemistry. In addition to its use in pharmaceuticals, trifluoroacetophenone is employed in the production of agrochemicals, including herbicides and insecticides. The trifluoromethyl group has been shown to enhance the effectiveness and persistence of agrochemicals, increasing their stability and providing greater control over target pests. The incorporation of trifluoromethyl groups in agrochemical molecules can also contribute to improved environmental resistance, making the active ingredients more durable in diverse weather conditions. Trifluoroacetophenone is also utilized in material science, particularly in the synthesis of specialty polymers and coatings. The presence of the trifluoromethyl group can improve the chemical and thermal stability of materials, as well as increase their resistance to solvents and other harsh environmental conditions. These properties make trifluoroacetophenone a useful precursor for creating high-performance materials for use in various industrial applications, such as electronics, coatings, and adhesives. Furthermore, trifluoroacetophenone has been explored for its potential in catalysis. Its ability to interact with metal centers due to the electron-withdrawing effects of the trifluoromethyl group can make it useful in catalytic processes, particularly in reactions such as the activation of certain reagents or the stabilization of reaction intermediates. This has been an area of ongoing research, particularly in the context of green chemistry and the development of more sustainable industrial processes. While trifluoroacetophenone is a valuable compound in various industrial and scientific applications, safety precautions must be observed when handling it. It is important to work with this chemical in well-ventilated areas and to use appropriate protective equipment, such as gloves and goggles, to avoid exposure. As with many fluorinated compounds, care should be taken to avoid inhalation or skin contact, as these can lead to irritation or other health concerns. In summary, trifluoroacetophenone is an important chemical intermediate with significant applications in the pharmaceutical, agrochemical, and material science industries. Its unique trifluoromethyl group imparts stability, reactivity, and bioactivity to the compound, making it a valuable building block for the development of a wide range of chemical products. Through its role in various chemical syntheses, trifluoroacetophenone continues to contribute to advancements in multiple fields of science and technology. References 1971. The EPR spectra of the anion radicals of trifluoroacetophenone, perfluorodimethylketene, and perfluoroacetone. Theoretical and Experimental Chemistry, 7(3). DOI: 10.1007/bf00523923 1968. Reactions of benzonitrile oxide with fluoroketones and their imines. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 17(8). DOI: 10.1007/bf01169917 1967. Reactions of polyfluoro ketones with aromatic compounds. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 16(3). DOI: 10.1007/bf00905999 |
| Market Analysis Reports |