Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Jinan Type Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.typechem.com | |||
 | +86 (531) 8806-2576 | |||
 | +86 (531) 8806-7257 | |||
 | xyudu@typechem.com xyudu@live.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Sigma-Aldrich, Inc. | China | |||
|---|---|---|---|---|
 | www.sigmaaldrich.com | |||
 | +86 (21) 6141-5566 800-819-3336 | |||
 | orderCN@sial.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Loop Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.looppharm.com | |||
 | +86 (0571) 8526-0761 | |||
 | sales01@looppharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Hangzhou Zentra Bio-Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zentrachem.com | |||
 | 18321999949 | |||
 | steven@zentrachem.com | |||
 | QQ Chat | |||
 | WeChat: Hangzhou Zentra Bio-Chemical Co., Ltd. | |||
| Chemical distributor since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic alcohol |
|---|---|
| Name | 3-Methylcatechol |
| Synonyms | 2,3-Dihydroxytoluene; 3-Methylbenzene-1,2-diol |
| Molecular Structure | 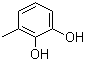 |
| Molecular Formula | C7H8O2 |
| Molecular Weight | 124.14 |
| CAS Registry Number | 488-17-5 |
| EC Number | 207-672-6 |
| SMILES | CC1=C(C(=CC=C1)O)O |
| Density | 1.2±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 65 - 68 °C (Expl.) |
| Boiling point | 240.9±20.0 °C 760 mmHg (Calc.)*, 241 °C (Expl.) |
| Flash point | 116.7±16.4 °C (Calc.)*, 140 °C (Expl.) |
| Solubility | water: soluble (Expl.) |
| Index of refraction | 1.595 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
3-Methylcatechol is an organic compound classified as a methyl-substituted derivative of catechol, also known as 1,2-dihydroxybenzene. Its structure consists of a benzene ring bearing two hydroxyl groups at the 1 and 2 positions (adjacent carbons), with a methyl group attached at the 3 position. This substitution pattern positions the methyl group ortho to one hydroxyl and meta to the other, which influences the compound’s chemical reactivity and physical properties. The compound is a colorless to pale yellow crystalline solid, soluble in water and common organic solvents such as ethanol and ether. Its melting point and solubility depend on purity and crystallization conditions. 3-Methylcatechol can be synthesized through various methods including methylation of catechol derivatives or hydroxylation of methyl-substituted benzenes. Biotechnological routes using microbial transformation of toluene or xylene derivatives also provide access to 3-methylcatechol, highlighting its relevance in environmental and industrial bioprocesses. Chemically, 3-methylcatechol exhibits properties typical of catechols, including susceptibility to oxidation and participation in redox reactions. The methyl substituent influences the electron density on the aromatic ring, modifying reactivity patterns compared to catechol. The two hydroxyl groups enable strong hydrogen bonding, affecting solubility and intermolecular interactions. 3-Methylcatechol finds applications primarily in organic synthesis as an intermediate for the preparation of pharmaceuticals, agrochemicals, and polymers. It serves as a precursor to compounds with antioxidant, antimicrobial, and UV-absorbing properties. The compound is also studied in environmental chemistry due to its formation during the biodegradation of aromatic hydrocarbons and its role as a substrate for enzymes such as catechol 1,2-dioxygenase. Analytical characterization of 3-methylcatechol includes nuclear magnetic resonance (¹H and ¹³C NMR) spectroscopy, which confirms the positions of hydroxyl and methyl substituents on the aromatic ring. Infrared (IR) spectroscopy detects characteristic hydroxyl group vibrations around 3400 cm–1 and aromatic C–H stretches. Mass spectrometry verifies molecular weight and fragmentation consistent with methylcatechol. In summary, 3-methylcatechol is a methyl-substituted catechol derivative with distinctive chemical properties due to its hydroxyl and methyl groups. It serves as an important intermediate in chemical synthesis and environmental processes, with well-documented methods of preparation and applications in various industrial and scientific fields. References 2024. 3-Methylcatechol mediates anti-fecundity effect by inhibiting estrogen-related receptor-induced glycolytic gene expression in Myzus persicae. Pesticide Biochemistry and Physiology, 200. DOI: 10.1016/j.pestbp.2024.105802 1968. Formation of 2-hydroxy-6-oxo-2, trans-4, trans-heptad-ienoic acid from 3-methylcatechol by a Pseudomonas. Cellular and Molecular Life Sciences, 24(2). DOI: 10.1007/bf02146927 |
| Market Analysis Reports |