Online Database of Chemicals from Around the World
| Beijing Huikang Boyuan Chemical Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.huikangchem.com | |||
 | +86 (10) 6886-2197 6886-7502 | |||
 | +86 (10) 6888-2204 | |||
 | market@huikangchem.com sales@huikangchem.com sales3@huikangchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2005 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Zehan Biopharma Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zehanbiopharma.com | |||
 | +86 (21) 6135-0663 +86 13052117465 | |||
 | +86 (21) 6135-0662 | |||
 | sales@zehanbiopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong Chemed Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemedpharma.com | |||
 | +86 (531) 8880-3416 | |||
 | +86 (531) 8880-3416 | |||
 | sales@chemedpharma.com | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Health Biochem Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.health-biochem.com | |||
 | +86 (29) 6259-9512 | |||
 | sales@health-biochem.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Nanjing Sunsure Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sunsurechem.com | |||
 | +86 (25) 5853-5435 | |||
 | +86 (25) 5853-5435 | |||
 | sales@sunsurechem.com | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Antiallergic >> Allergic reaction medium blocker |
|---|---|
| Name | Ibudilast |
| Synonyms | 3-Isobutyryl-2-isopropylpyrazolo[1,5-a]pyridine; 2-Methyl-1-[2-(1-methylethyl)pyrazolo[1,5-a]pyridin-3-yl]-1-propanone; 2-Isopropyl-3-isobutyrylpyrazolo[1,5-a]pyridine |
| Molecular Structure | 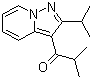 |
| Molecular Formula | C14H18N2O |
| Molecular Weight | 230.31 |
| CAS Registry Number | 50847-11-5 |
| EC Number | 637-150-7 |
| SMILES | CC(C)C1=NN2C=CC=CC2=C1C(=O)C(C)C |
| Density | 1.1±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 56 °C (Expl.) |
| Solubility | Soluble 100 mM (DMSO), 100 mM (ethanol) (Expl.) |
| Index of refraction | 1.571 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
Ibudilast is a small-molecule pharmaceutical compound known primarily for its action as a non-selective phosphodiesterase (PDE) inhibitor and as a modulator of glial cell activity. It is chemically classified as a benzofuran derivative and is used in several countries, including Japan and South Korea, for the treatment of asthma and post-stroke complications. Its structure includes a benzofuran ring substituted with an amide and alkyl groups that contribute to its lipophilicity and oral bioavailability. The compound was first developed in Japan during the 1970s by Nippon Kayaku Co., Ltd. as part of a broader research effort into anti-inflammatory agents targeting cyclic nucleotide metabolism. Ibudilast's inhibition of PDE enzymes increases intracellular levels of cyclic AMP (cAMP) and cyclic GMP (cGMP), which in turn mediate smooth muscle relaxation, bronchodilation, and modulation of inflammatory responses. Its activity is not limited to a single PDE isoform; rather, it exhibits moderate inhibition across several isoforms including PDE3, PDE4, PDE10, and PDE11. In Japan, ibudilast has been approved under the trade name Ketas for the treatment of bronchial asthma, chronic cerebral circulation disorders, and as a preventive agent for secondary cerebral infarction. It improves blood flow by inhibiting platelet aggregation and promoting vasodilation, and its neuroprotective and anti-inflammatory properties are considered beneficial in the post-stroke rehabilitation setting. Its ability to inhibit glial cell activation also contributes to its neuroprotective potential by suppressing the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6. Beyond its approved uses, ibudilast has attracted international research attention for its potential therapeutic applications in neuropathic pain, multiple sclerosis (MS), opioid withdrawal, and substance use disorders. Studies have shown that ibudilast can reduce opioid tolerance and dependence by inhibiting glial activation and suppressing neuroinflammation, mechanisms that differ from traditional opioid antagonists. It has been investigated in the United States under investigational new drug (IND) applications and orphan drug designations for conditions including progressive MS and opioid use disorder. Ibudilast is typically administered orally in tablet or capsule form. It is rapidly absorbed and undergoes hepatic metabolism, with its pharmacokinetics influenced by enterohepatic circulation. The drug has a relatively long half-life, allowing for twice-daily dosing in clinical settings. Common side effects reported during its use include nausea, headache, and abdominal discomfort, though it is generally well tolerated. Analytical methods for characterizing and quantifying ibudilast in pharmaceutical preparations include high-performance liquid chromatography (HPLC), mass spectrometry (MS), and infrared (IR) spectroscopy. The compound is a pale-yellow crystalline solid, sparingly soluble in water but readily soluble in organic solvents such as ethanol and methanol. Stability studies indicate that ibudilast is stable under standard storage conditions but should be protected from excessive heat and light. In recent years, the compound has also been investigated for its potential use in treating amyotrophic lateral sclerosis (ALS), Alzheimer's disease, and other neurodegenerative conditions. These studies are ongoing, and while ibudilast has not yet received regulatory approval in most Western countries, clinical trials continue to evaluate its efficacy and safety in diverse therapeutic areas. In summary, ibudilast is a well-established phosphodiesterase inhibitor and glial modulator originally developed in Japan for asthma and cerebral circulation disorders. Its unique pharmacological profile has made it a subject of active investigation in neurology and addiction medicine. Its combination of anti-inflammatory, neuroprotective, and vasodilatory actions positions it as a candidate for expanded use in treating central nervous system disorders and inflammatory conditions. References 2024. Targeting phosphodiesterase 4 as a potential therapy for Parkinson’s disease: a review. Molecular Biology Reports, 51(5). DOI: 10.1007/s11033-024-09484-8 2024. Molecular Indicator for Distinguishing Multi-drug-Resistant Tuberculosis from Drug Sensitivity Tuberculosis and Potential Medications for Treatment. Molecular Biotechnology. DOI: 10.1007/s12033-024-01299-z 2024. Morphine self-administration is inhibited by the antioxidant N-acetylcysteine and the anti-inflammatory ibudilast; an effect enhanced by their co-administration. PLOS ONE, 19(10). DOI: 10.1371/journal.pone.0312828 |
| Market Analysis Reports |