Deoxynivalenol is a naturally occurring mycotoxin produced primarily by certain species of Fusarium fungi, especially Fusarium graminearum and Fusarium culmorum. It belongs to the trichothecene family of sesquiterpenoid toxins and is also known as vomitoxin due to its emetic effects observed in animals. Deoxynivalenol commonly contaminates cereal crops such as wheat, barley, oats, and maize, posing significant risks to food safety and agricultural production worldwide.
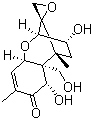
The discovery of deoxynivalenol dates back to the mid-20th century when researchers identified it as a major toxic metabolite associated with Fusarium head blight, a devastating fungal disease affecting cereal crops. Since then, its chemical structure and toxicological properties have been extensively studied. Deoxynivalenol has a tetracyclic 12,13-epoxytrichothec-9-ene skeleton, which is characteristic of type B trichothecenes. The presence of an epoxide group is critical to its biological activity and toxicity.
Deoxynivalenol acts by inhibiting protein synthesis in eukaryotic cells. It binds to the ribosomal peptidyl transferase center, disrupting the elongation phase of translation and leading to cellular stress responses. This mechanism underlies many of its toxic effects, including immunosuppression, gastrointestinal disturbances, and interference with cell proliferation. The toxin can induce apoptosis and inflammation, contributing to its overall toxicity profile.
In agriculture and food safety, deoxynivalenol is a major concern due to its prevalence in contaminated grains. It compromises crop quality and yield, and when ingested by humans or animals, it can cause acute and chronic health effects. Regulatory agencies worldwide have established maximum allowable limits for deoxynivalenol levels in food and feed to protect consumer health. Detection and quantification methods, including chromatographic and immunoassay techniques, have been developed to monitor contamination levels in agricultural products.
Deoxynivalenol contamination poses particular risks to livestock, especially swine, which are highly sensitive to its toxic effects. Exposure can result in feed refusal, vomiting, reduced weight gain, and impaired immune function. In humans, consumption of contaminated food can lead to nausea, vomiting, diarrhea, and other gastrointestinal symptoms. Chronic exposure has also been associated with immunotoxicity and potential impacts on growth and development.
Research into deoxynivalenol has also focused on strategies to mitigate its impact. These include breeding crop varieties resistant to Fusarium infection, employing agricultural practices to reduce fungal growth, and using detoxification agents or adsorbents in animal feed to limit toxin bioavailability. Advances in molecular biology have improved understanding of fungal toxin biosynthesis pathways, enabling the development of targeted interventions.
Overall, deoxynivalenol represents a significant challenge in food safety and agricultural management. Its well-characterized toxicological mechanisms and widespread occurrence have driven extensive research to detect, control, and mitigate its presence in the food chain. Continued efforts in monitoring, crop protection, and risk assessment are essential to minimize the health risks associated with this mycotoxin.
References
1984. Effect of deoxynivalenol (vomitoxin) on the humoral IM-munity of mice. Toxicology Letters, 23(2-3).
DOI: 10.1016/0378-4274(84)90004-3
2008. Wheat-infecting Fusarium species in Poland — their chemotypes and frequencies revealed by PCR assay. Journal of Applied Genetics, 49(4).
DOI: 10.1007/bf03195644
2009. Deoxynivalenol-induced cytotoxicity, cytokines and related genes in unstimulated or lipopolysaccharide stimulated primary porcine macrophages. Toxicology Letters, 184(2).
DOI: 10.1016/j.toxlet.2008.10.024
|



































 GHS06 Danger Details
GHS06 Danger Details