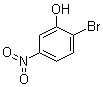
2‑Bromo‑5‑nitrophenol (C6H4BrNO3) is an aromatic nitro compound substituted with a bromine atom at the 2‑position and a nitro group at the 5‑position of the phenol ring. It is primarily used as a synthetic intermediate in organic chemistry rather than being a biologically active molecule itself. Its chemical structure, containing both an electron‑withdrawing nitro group and a bromine atom, imparts reactivity that is exploited in multistep organic syntheses.
The phenolic hydroxyl group can undergo derivatization through protection or etherification, allowing selective reactions at other positions of the aromatic ring. The nitro group can be chemically reduced to an amino group, forming 2‑bromo‑5‑aminophenol, which serves as a versatile intermediate for the construction of heterocyclic compounds, dyes, pharmaceuticals, and other functional organic molecules. The bromine atom is useful in palladium‑catalyzed cross‑coupling reactions such as Suzuki or Buchwald–Hartwig couplings, enabling replacement of the bromine with carbon or heteroatom substituents to generate complex aryl systems.
2‑Bromo‑5‑nitrophenol’s applications are largely in the pharmaceutical and fine chemical industries, where it serves as a building block for active pharmaceutical ingredients or other specialty chemicals. It is also used in material science research as a precursor for the synthesis of functional molecules in organic electronics or sensors. The combination of nitro, phenol, and bromine functionalities makes it a flexible starting material for various transformations, including reductions, substitutions, and cross-coupling reactions.
Handling of 2‑Bromo‑5‑nitrophenol requires appropriate safety precautions due to its nitroaromatic character. It is harmful if ingested, can cause skin and eye irritation, and should be stored under cool, dry conditions. Its stability and reactivity make it convenient for commercial distribution as a catalog chemical, widely available for laboratory synthesis purposes.
References
Kashihara M, Sato S, Tobisu M (2021) Cross‑coupling reactions of nitroarenes. Accounts of Chemical Research 54(22): … DOI: 10.1021/acs.accounts.1c00220
Emadi R, Rostamnia S, Esmaeilpour M (2023) Applications of palladium‑catalyzed C–N cross‑coupling in pharmaceutical compound synthesis. RSC Advances DOI: 10.1039/D2RA07412E
Crawley ML, Walsh PJ (2012) Applications of transition‑metal catalysis in drug discovery and development. In: Catalysis in Organic Synthesis – An Industrial Perspective. Link
|

















 GHS09 Warning Details
GHS09 Warning Details