Online Database of Chemicals from Around the World
| Yicheng Goto Pharmaceuticals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gotopharm.com | |||
 | +86 (710) 342-3122 342-3123 | |||
 | +86 (710) 342-3124 | |||
 | info@gotopharm.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hubei Gedian Humanwell Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.steroid-chem.com | |||
 | +86 (27) 8759-7155 | |||
 | +86 (27) 8759-7383 | |||
 | market@steroid-chem.com | |||
| Chemical manufacturer since 2000 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Kewel Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kewelchem.com | |||
 | +86 (21) 6460-9169 | |||
 | +86 (21) 6460-9160 | |||
 | kewelchem88@gmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhejiang Xianju Junye Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.junyepharm.com | |||
 | +86 (576) 8782-1302 | |||
 | junye@junyepharm.com 16155972@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Qichuang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qc-chemical.com | |||
 | +86 (571) 8893-5129 | |||
 | +86 (571) 8825-0182 | |||
 | david@qc-chemical.com sales@qc-chemical.com davidw0828@gmail.com | |||
 | QQ Chat | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Baoding Jiahe Fine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bdchem.com | |||
 | +86 (312) 209-7097 209-7095 | |||
 | +86 (312) 209-7096 | |||
 | sales@jiahe-pharm.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Eastar Chemical Corporation | USA | |||
|---|---|---|---|---|
 | www.eastarchem.com | |||
 | +1 800-898-2436 | |||
 | +1 (877) 898-2436 | |||
 | info@eastarchem.com | |||
| Chemical manufacturer since 1989 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Shilo Pharmachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shilopharma.com | |||
 | +86 15258872085 | |||
 | sales@shilopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Chengdu Biopurify Phytochemicals Ltd. | China | |||
|---|---|---|---|---|
 | www.phytopurify.com | |||
 | +86 (28) 8263-3860 8263-3987 | |||
 | +86 (28) 8263-3165 | |||
 | sales@biopurify.com biopurify@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2017 | ||||
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Hebei Ganmiao New Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hbganmiao.com | |||
 | +86 13897677193 | |||
 | sales2@hbganmiao.com | |||
 | WeChat: 13897677193 | |||
 | WhatsApp:+8613897677193 | |||
| Chemical distributor since 2024 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Kingchem Inc. | USA | |||
|---|---|---|---|---|
 | www.kingchem.com | |||
 | +1 (800) 211-4330 (201) 825-9988 | |||
 | +1 (201) 825-9148 | |||
 | customer-service@kingchem.com | |||
| Chemical manufacturer since 1994 | ||||
| Xi'an Gaoyuan Bio-Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gaoyuanbio.cn | |||
 | +86 (29) 8209-8892 8209-8893 | |||
 | +86 (29) 8209-8893 | |||
 | puyu@gaoyuanbio.com | |||
| Chemical manufacturer | ||||
| CarboMer, Inc. | USA | |||
|---|---|---|---|---|
 | www.carbomer.com | |||
 | +1 (858) 552-0992 | |||
 | +1 (858) 552-0999 | |||
 | sales@carbomer.com | |||
| Chemical manufacturer | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| W & J PharmaChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.wjpharmachem.com | |||
 | +1 (301) 880-0624 | |||
 | +1 (301) 434-9366 | |||
 | info@wjpharmachem.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| ECA International Corporation | USA | |||
|---|---|---|---|---|
 | www.ecacorporation.com | |||
 | +1 (847) 358-8178 | |||
 | +1 (847) 358-8179 | |||
 | order@ecacorporation.com | |||
| Chemical manufacturer | ||||
| City Chemical LLC | USA | |||
|---|---|---|---|---|
 | www.citychemical.com | |||
 | +1 (203) 932-2489 | |||
 | +1 (203) 937-8400 | |||
 | sales@citychemical.com | |||
| Chemical distributor | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Methyl pyrimidine |
|---|---|
| Name | Dehydroepiandrosterone |
| Synonyms | DHEA; Prasterone; Androstenolone; Dehydroisoandrosterone; trans-Dehydroandrosterone; Hydroxyandrost-5-en-17-one |
| Molecular Structure | 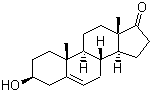 |
| Molecular Formula | C19H28O2 |
| Molecular Weight | 288.43 |
| CAS Registry Number | 53-43-0 |
| EC Number | 200-175-5 |
| SMILES | C[C@]12CC[C@H]3[C@H]([C@@H]1CCC2=O)CC=C4[C@@]3(CC[C@@H](C4)O)C |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 1151 °C |
| alpha | 12 ° (c=2, ethanol 96% 25 °C) |
| Index of Refraction | 1.560, Calc.* |
| Boiling Point | 426.7±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 182.1±21.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Danger Details GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H351-H360-H362-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P260-P261-P263-P264-P264+P265-P270-P271-P273-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P318-P319-P321-P330-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||
|
Dehydroepiandrosterone, commonly known as DHEA, is a naturally occurring steroid hormone synthesized primarily in the adrenal glands. Its chemical formula is C19H28O2, and it serves as a precursor to both androgens and estrogens. DHEA was first isolated and identified in the 1930s, with significant advancements in its understanding occurring over the subsequent decades. Researchers recognized its role in steroidogenesis, the process by which steroids are produced from cholesterol, establishing DHEA as a crucial player in human hormonal balance. The discovery of DHEA is attributed to the work of several scientists in the early 20th century, who were investigating the chemistry of adrenal hormones. The compound was first isolated in 1934 by the biochemist Adolf Butenandt, who identified it as a significant component of human and animal adrenal extracts. The term "dehydroepiandrosterone" reflects its chemical structure, indicating that it is derived from pregnenolone and is an androgenic hormone. Throughout the mid-20th century, further research confirmed its various biological roles, particularly in the regulation of metabolism, immune function, and sexual health. DHEA serves multiple functions in the human body, primarily acting as a precursor to other hormones. It is converted into testosterone and estrogen in peripheral tissues, thus playing a vital role in the development of secondary sexual characteristics, reproductive health, and overall hormonal balance. DHEA levels peak in early adulthood and gradually decline with age, leading to interest in its supplementation as a means to counteract age-related hormonal decline. One of the primary applications of DHEA is in the field of hormone replacement therapy. Supplementation with DHEA has been proposed as a method to restore hormonal levels in aging individuals, particularly those experiencing symptoms associated with low levels of androgens and estrogens, such as fatigue, depression, and decreased libido. Clinical studies have suggested that DHEA supplementation may improve mood, enhance cognitive function, and promote overall well-being in some populations, although the results remain mixed and more research is needed to establish definitive benefits. In addition to its role in hormone therapy, DHEA has been investigated for its potential applications in enhancing athletic performance and muscle strength. Some athletes and bodybuilders use DHEA as a performance-enhancing supplement, as it may aid in increasing muscle mass and improving physical endurance. However, its use in sports is controversial, and regulatory bodies such as the World Anti-Doping Agency (WADA) have banned DHEA in competitive sports due to concerns about its performance-enhancing effects and potential side effects. DHEA has also been studied for its potential role in various health conditions, including osteoporosis, cardiovascular disease, and metabolic disorders. Some research indicates that DHEA supplementation may enhance bone density and improve insulin sensitivity, although results vary among individuals. Furthermore, DHEA is being explored for its potential neuroprotective effects, with preliminary studies suggesting it may have a role in preventing cognitive decline associated with aging and neurodegenerative diseases. Despite the interest in DHEA supplementation, safety and efficacy remain subjects of ongoing research. Side effects associated with DHEA use can include hormonal imbalances, acne, hair loss, and changes in mood. As a result, individuals considering DHEA supplementation should consult healthcare professionals to assess their specific needs and potential risks. In summary, dehydroepiandrosterone is a significant steroid hormone with a complex history and diverse applications in health and wellness. Its discovery has contributed to our understanding of hormonal balance and the role of androgens and estrogens in human physiology. Ongoing research continues to explore its potential benefits and applications, underscoring its importance in the fields of endocrinology and age-related health. References 1990. Dehydroepiandrosterone and a beta-agonist, energy transducers, alter antioxidant enzyme systems: influence of chronic training and acute exercise in rats. Archives of Biochemistry and Biophysics. DOI: 10.1016/0003-9861(90)90674-n 1994. Specific binding of dehydroepiandrosterone sulfate to rat liver cytosol: A possible association with peroxisomal enzyme induction. Biochimica et Biophysica Acta (BBA) - Molecular Cell Research. DOI: 10.1016/0167-4889(94)90121-x 1998. Oxygen free radical scavenger properties of dehydroepiandrosterone. Cell Biochemistry and Function. DOI: 10.1002/(sici)1099-0844(199803)16:1<57::aid-cbf771>3.0.co;2-s |
| Market Analysis Reports |