Online Database of Chemicals from Around the World
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Organic derivative of hydrazine or hydrazine |
|---|---|
| Name | Methyldithiocarbazate |
| Synonyms | methyl N-aminocarbamodithioate |
| Molecular Structure | 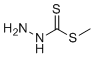 |
| Molecular Formula | C2H6N2S2 |
| Molecular Weight | 122.21 |
| CAS Registry Number | 5397-03-5 |
| EC Number | 812-150-1 |
| SMILES | CSC(=S)NN |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.645, Calc.* |
| Boiling Point | 207.3±23.0 °C (760 mmHg), Calc.* |
| Flash Point | 79.2±22.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Methyldithiocarbazate is an organic compound with a chemical structure that includes a dithiocarbazate group and a methyl group. It is primarily used in synthetic chemistry, particularly in the preparation of metal coordination complexes and as a reagent in various chemical reactions. The compound has garnered attention in diverse scientific fields, including medicinal chemistry, material science, and environmental chemistry, due to its unique chemical properties and versatility in different applications. Methyldithiocarbazate was first synthesized and characterized in the mid-20th century as part of ongoing research into dithiocarbazate derivatives. The dithiocarbazate group, consisting of a sulfur-nitrogen bond, is known for its reactivity and ability to form stable complexes with metals, which makes it useful in a variety of chemical processes. The addition of a methyl group to the structure enhances its solubility and stability, expanding its potential applications. The most notable application of methyldithiocarbazate is in the field of coordination chemistry. The compound is commonly used as a ligand in the synthesis of metal complexes. It has the ability to bind to metal ions such as copper, zinc, and iron, forming coordination complexes that exhibit unique properties. These complexes have potential applications in catalysis, electrochemistry, and as functional materials in various industries. Methyldithiocarbazate-derived metal complexes have also been investigated for their antimicrobial properties, showing promise as candidates for the development of new antimicrobial agents. In addition to its use in coordination chemistry, methyldithiocarbazate has applications in medicinal chemistry. Some studies have indicated that it may possess biological activities, such as anti-inflammatory and anticancer effects. Research has also explored its potential as a chelating agent for heavy metal ions, particularly in the context of environmental remediation. The ability of methyldithiocarbazate to bind to and remove toxic metal ions from contaminated environments could make it a valuable tool in addressing environmental pollution, especially in areas affected by industrial waste or mining operations. Furthermore, methyldithiocarbazate has been investigated for its potential in the synthesis of novel materials, including organic semiconductors and sensors. Its ability to form stable complexes with various metals makes it useful in the development of new electronic devices and materials with tailored properties. Researchers are particularly interested in its potential to contribute to the development of environmentally friendly materials and processes. Despite its promising applications, there is ongoing research to better understand the full range of properties and potential uses of methyldithiocarbazate. Studies continue to explore its reactivity, toxicity, and environmental impact, particularly in the context of long-term exposure and use in industrial and environmental applications. As research progresses, methyldithiocarbazate may find further applications in fields such as drug development, materials science, and environmental engineering. References 1978. Some transition metal complexes of the ligand methyldithiocarbazate. Australian Journal of Chemistry, 31(6). DOI: 10.1071/CH9781347 2012. Synthesis and biological evaluation of some new thiadiazole derivatives. Medicinal Chemistry Research, 21(9). DOI: 10.1007/s00044-011-9778-0 2011. Synthesis, characterization and biological activity of Mn(II), Co(II), Ni(II), Cu(II) and Zn(II) complexes of a Schiff base derived from methyldithiocarbazate. Transition Metal Chemistry, 36(3). DOI: 10.1007/s11243-011-9466-3 |
| Market Analysis Reports |