Dibutyl sulfide is an organosulfur compound consisting of two n-butyl groups linked by a sulfur atom. Its molecular formula is C8H18S. The compound appears as a colorless to pale yellow liquid under standard conditions, with a characteristic odor typical of sulfides. Dibutyl sulfide is moderately soluble in nonpolar organic solvents such as hexane, benzene, and chloroform, while being poorly soluble in water due to its hydrophobic hydrocarbon chains.
The discovery and use of dibutyl sulfide are part of the broader study of dialkyl sulfides, which have been investigated for their chemical reactivity, industrial applications, and role as intermediates in organic synthesis. Sulfides like dibutyl sulfide are notable for their nucleophilic sulfur atom, which can participate in substitution reactions, oxidation, and coordination to transition metals, making them versatile building blocks in chemical processes.
Synthesis of dibutyl sulfide is commonly achieved through nucleophilic substitution reactions. One typical method involves the reaction of n-butyl halides (such as 1-bromobutane or 1-chlorobutane) with sodium sulfide (Na2S) or other sulfide salts in an appropriate solvent under controlled temperature. The reaction proceeds via a bimolecular nucleophilic substitution mechanism, resulting in the formation of the symmetric sulfide. Purification is typically performed by distillation to remove unreacted starting materials and side products.
Chemically, dibutyl sulfide exhibits the characteristic reactivity of thioethers. The sulfur atom, being more nucleophilic than oxygen in analogous ethers, can undergo oxidation to form sulfoxides or sulfones. It can also act as a ligand for transition metals, forming coordination complexes that are useful in catalysis and organometallic chemistry. The alkyl chains provide steric bulk and hydrophobicity, which influence solubility, boiling point, and reactivity.
In practical applications, dibutyl sulfide is used as an intermediate in the synthesis of pharmaceuticals, agrochemicals, and specialty chemicals. It serves as a precursor to sulfoxides and sulfones, which are commonly employed in medicinal chemistry for drug development and in materials science for polymer modification. Additionally, dibutyl sulfide can function as a solvent or additive in chemical reactions requiring a nucleophilic sulfur source or a hydrophobic medium.
From a physical perspective, dibutyl sulfide is stable under ambient conditions but should be protected from strong oxidizing agents that can convert it to sulfoxides or sulfones. It is flammable, and adequate safety precautions, including the use of gloves, goggles, and proper ventilation, are necessary during handling and storage. Its characteristic odor can also pose handling challenges, requiring containment in well-ventilated environments.
Overall, dibutyl sulfide is a chemically versatile organosulfur compound. Its combination of nucleophilic sulfur, hydrophobic alkyl chains, and moderate stability makes it a valuable intermediate in organic synthesis and industrial chemistry. The compound’s ability to undergo oxidation, substitution, and coordination reactions underpins its broad utility in chemical research, materials development, and the production of functionalized sulfur-containing molecules.
References
2025. Study of Ligands on the Oxygenation Curve and Oxidation Rate of Heme Dimethyl Ester Hexacoordinated Complexes. Russian Journal of General Chemistry.
DOI: 10.1134/s1070363225601449
|











































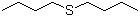
 GHS07 Warning Details
GHS07 Warning Details