Online Database of Chemicals from Around the World
| Nanjing Search Biotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.searchbio.com.cn | |||
 | +86 (25) 8168-2922 8586-0978 +86 18913919581 | |||
 | +86 (25) 8586-0962 | |||
 | trade@searchbio.com.cn sales@searchbio.com.cn Linda@searchbio.com.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Pure Chemistry Scientific Inc | USA | |||
|---|---|---|---|---|
 | www.purechems.com | |||
 | +1 (888) 588-9418 | |||
 | +1 (617) 206-9595 | |||
 | sales@purechems.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Biochempeg | USA | |||
|---|---|---|---|---|
 | www.biochempeg.com | |||
 | +1 (857) 366-6766 | |||
 | +1 (617) 206-9595 | |||
 | sales@biochempeg.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Dalo Chem Life Sciences (Dongguan) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.dalochem.com | |||
 | +86 15019076757 | |||
 | sales@dalochem.com | |||
 | WhatsApp:+86-15019076757 | |||
| Chemical distributor since 2023 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Kishida Chemical Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.kishida.co.jp | |||
 | +81 (6) 6946-8061 | |||
 | +81 (6) 6946-1607 | |||
 | shiyaku@kishida.co.jp | |||
| Chemical manufacturer | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Advance Research Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.fluoridearc.com | |||
 | +1 (918) 266-6789 | |||
 | +1 (918) 266-6796 | |||
 | sales@fluoridearc.com | |||
| Chemical manufacturer | ||||
| Indofine Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.indofinechemical.com | |||
 | +1 (888) 463-6346 | |||
 | +1 (908) 359-1179 | |||
 | info@indofinechemical.com | |||
| Chemical manufacturer since 1981 | ||||
| Suzhou BEC Fine Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bek.com.cn | |||
 | +86 (512) 6809-5917 +86 13806207098 | |||
 | +86 (512) 6841-7933 | |||
 | bek@bek.com.cn | |||
| Chemical manufacturer | ||||
| Santa Cruz Biotechnology, Inc. | USA | |||
|---|---|---|---|---|
 | www.scbt.com | |||
 | +1 (831) 457-3800 | |||
 | +1 (831) 457-3801 | |||
 | scbt@scbt.com | |||
| Chemical manufacturer | ||||
| Zibo Xingzhilian Chemistry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xingzhilian.com | |||
 | +86 (311) 8201-6547 8201-2958 | |||
 | +86 (311) 8201-7132 | |||
 | zbfetdc@zb-public.sd.cninfo.net | |||
| Chemical manufacturer since 2001 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Piperazine |
|---|---|
| Name | PIPES |
| Synonyms | 1,4-Piperazinebis(ethanesulfonic acid); Piperazine-1,4-bis(2-ethanesulfonic acid); Piperazine-N,N'-bis(2-ethanesulfonic acid) |
| Molecular Structure | 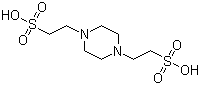 |
| Molecular Formula | C8H18N2O6S2 |
| Molecular Weight | 302.36 |
| CAS Registry Number | 5625-37-6 |
| EC Number | 227-057-6 |
| SMILES | C1CN(CCN1CCS(=O)(=O)O)CCS(=O)(=O)O |
| Melting point | 300 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P305+P351+P338 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
PIPES (piperazine-N,N'-bis(2-ethanesulfonic acid)) is a zwitterionic buffer discovered by Norman E. Good and co-workers in the 1960s. The development of PIPES was part of a broader effort to create Good's buffers, which maintain stable pH in biochemical and biological studies, answering the need for nontoxic, stable, and minimally interacting substances that could reliably maintain pH under a wide range of experimental conditions. The molecular formula of PIPES is C8H18N2O6S2, with a molecular weight of 302.37 g/mol. It features a piperazine ring attached to two ethanesulfonic acid groups. This unique structure gives PIPES high buffering capacity and stability, especially in the pH range of 6.1 to 7.5, with a pKa of 6.8 at 25°C. These properties make PIPES particularly useful for maintaining near-neutral pH in biological and biochemical experiments. PIPES is widely used in cell culture media to maintain pH within the physiological range. Its nontoxic nature ensures that it does not interfere with cell metabolism or viability, making it an ideal buffer for culturing and maintaining a variety of cell types under controlled conditions. PIPES is a popular electrophoresis buffer, particularly in the separation of proteins and nucleic acids. It is commonly used in polyacrylamide gel electrophoresis (PAGE) and agarose gel electrophoresis. The stability of PIPES at near-neutral pH helps ensure accurate migration and separation of macromolecules, resulting in clear and reproducible results. In biochemical analysis, PIPES is used to stabilize pH, which is critical for the activity and stability of enzymes and other biomolecules. Its buffering capacity ensures that analytical conditions remain optimal, allowing for accurate and reliable measurements of biochemical activity. PIPES is used in protein purification protocols to maintain stable pH conditions, which is critical for preserving protein structure and function. Its compatibility with a variety of chromatographic techniques makes it a valuable buffer in protein purification and analysis. In structural biology, PIPES is used in crystallography and other techniques to study the structure of biomolecules. Its ability to maintain stable pH conditions without interacting with the biomolecules being studied is essential for obtaining high-quality structural data. The high buffering capacity makes PIPES effective in maintaining a stable pH, ensuring consistent experimental conditions. The low ionic strength makes PIPES compatible with a wide range of biological systems and techniques, minimizing interference with the process being studied. PIPES is generally nontoxic to cells and does not interfere with biochemical reactions, making it suitable for use in sensitive biological applications. References 2024. Chelation of actinium with D-peptide linkers. Nature Communications, 15(1). DOI: 10.1038/s41467-024-50017-5 2024. Structural basis of microtubule stabilization by tubulin acetyltransferase. Nature Structural & Molecular Biology, 31(4). DOI: 10.1038/s41594-024-01406-3 1975. Sodium uptake in Anacystis nidulans. Zeitschrift für Pflanzenphysiologie, 76(3). DOI: 10.1007/bf00492037 |
| Market Analysis Reports |