Online Database of Chemicals from Around the World
| Tianjin Pharmacn Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharmacn.com | |||
 | +86 (22) 8372-6121 | |||
 | +86 (22) 2337-7103 | |||
 | marketing@pharmacn.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Shanghai Tajilin Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tajilin.com | |||
 | +86 (21) 5063-0626 | |||
 | +86 (21) 5056-3898 | |||
 | sales003@tajilin.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| Chess GmbH | Germany | |||
|---|---|---|---|---|
 | www.chess-chem.com | |||
 | +49 (621) 318-9794 | |||
 | +49 (621) 318-9691 | |||
 | sales@chess-chem.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Piperazine |
|---|---|
| Name | 1-(Cyclopropylcarbonyl)piperazine |
| Synonyms | cyclopropyl(piperazin-1-yl)methanone |
| Molecular Structure | 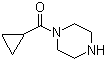 |
| Molecular Formula | C8H14N2O |
| Molecular Weight | 154.21 |
| CAS Registry Number | 59878-57-8 |
| EC Number | 626-499-0 |
| SMILES | C1CC1C(=O)N2CCNCC2 |
| Density | 1.1$+/-$0.1 g/cm3 Calc.*, 1.087 g/mL (Expl.) |
|---|---|
| Boiling point | 305.5$+/-$35.0 $degree$C 760 mmHg (Calc.)*, 426.3 $degree$C (Expl.) |
| Flash point | 138.5$+/-$25.9 $degree$C (Calc.)*, 110 $degree$C (Expl.) |
| Index of refraction | 1.535 (Calc.)*, 1.524 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H318-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P354+P338-P317-P319-P321-P332+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
1-(Cyclopropylcarbonyl)piperazine is a heterocyclic compound in which a piperazine ring is substituted at the nitrogen with a cyclopropylcarbonyl group. Piperazine derivatives are commonly used in medicinal chemistry because of their conformational flexibility, basicity, and ability to form hydrogen bonds with biological targets. The cyclopropylcarbonyl group introduces both steric hindrance and electronic modulation, which can influence metabolic stability, lipophilicity, and receptor binding, making this compound a versatile intermediate for drug discovery and synthetic applications. Structurally, the molecule consists of a six-membered piperazine ring with nitrogen atoms at positions 1 and 4. The N1 nitrogen is acylated with a cyclopropylcarbonyl moiety, forming an amide linkage (–NH–CO–cyclopropyl). The cyclopropyl group is a strained three-membered ring that can impart unique steric and electronic properties to the molecule, influencing interactions with enzymes or receptors. The secondary nitrogen in the piperazine ring remains available for further functionalization, allowing the compound to serve as a bifunctional building block. The synthesis of 1-(cyclopropylcarbonyl)piperazine typically involves the reaction of piperazine with cyclopropanecarbonyl chloride or cyclopropanecarboxylic acid derivatives under conditions that promote selective acylation of one nitrogen. Reagents such as bases or coupling agents are employed to facilitate the formation of the amide bond while minimizing diacylation of both nitrogen atoms. This selective acylation produces a stable intermediate that can be used for further chemical modifications or incorporated into more complex heterocyclic systems. In medicinal chemistry, this compound is valuable as an intermediate for designing drugs that target central nervous system receptors, transporters, or enzymes. The piperazine ring provides aqueous solubility and conformational flexibility, while the cyclopropylcarbonyl group can enhance binding specificity and metabolic stability. The combination of a secondary nitrogen and an acylated nitrogen allows chemists to selectively introduce additional substituents, such as alkyl, aryl, or heteroaryl groups, enabling the generation of compound libraries for structure–activity relationship studies. Beyond pharmacological applications, 1-(cyclopropylcarbonyl)piperazine is a useful synthetic intermediate in organic chemistry. The free nitrogen in the piperazine can undergo further derivatization, including alkylation, acylation, or sulfonylation, while the amide bond provides a stable handle for incorporation into larger molecules. Its combination of a strained cyclopropyl ring and heterocyclic piperazine core makes it suitable for the construction of bioactive molecules, heterocyclic analogs, and functionalized chemical libraries. Overall, 1-(cyclopropylcarbonyl)piperazine exemplifies a functionalized piperazine derivative with a secondary amide and a free nitrogen. Its structural features provide chemical versatility, stereoelectronic modulation, and applicability in medicinal chemistry and organic synthesis, making it a valuable building block for the development of bioactive compounds and heterocyclic intermediates. References 2023. Open science discovery of potent noncovalent SARS-CoV-2 main protease inhibitors. Science (New York, N.Y.). DOI: 10.1126/science.abo7201 2021. Improving the reaction efficiency of condensation amidation of piperazine with benzoic acid based on kinetics study in microreactors. Journal of Flow Chemistry. DOI: 10.1007/s41981-021-00166-1 2015. Olaparib. Pharmaceutical Substances. |
| Market Analysis Reports |