Online Database of Chemicals from Around the World
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Sinocure Chemical Group | China | |||
|---|---|---|---|---|
 | www.sinocurechem.com | |||
 | +86 15550440621 | |||
 | info@sinocurechem.com | |||
| Chemical manufacturer since 2020 | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Aromatic ketone |
|---|---|
| Name | 2,2-Diethoxyacetophenone |
| Synonyms | 2,2-Diethoxy-1-phenylethanone; DEAP |
| Molecular Structure | 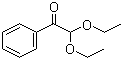 |
| Molecular Formula | C12H16O3 |
| Molecular Weight | 208.26 |
| CAS Registry Number | 6175-45-7 |
| EC Number | 228-220-4 |
| SMILES | CCOC(C(=O)C1=CC=CC=C1)OCC |
| Density | 1.034 |
|---|---|
| Boiling point | 131-134 °C (10 mmHg) |
| Refractive index | 1.4985-1.5005 |
| Flash point | 128 °C |
| Water solubility | 0.16 g/L (25 °C) |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H319-H335-H351 Details | ||||||||||||||||||||
| Safety Statements | P203-P261-P264+P265-P271-P280-P304+P340-P305+P351+P338-P318-P319-P337+P317-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2,2-Diethoxyacetophenone is an important compound in organic chemistry, recognized primarily for its utility as a photoinitiator in polymerization processes. This substance, characterized by its acetophenone core and two ethoxy groups, has been integral to advancements in UV-curable materials. The discovery of 2,2-diethoxyacetophenone can be traced back to the mid-20th century, as researchers sought effective photoinitiators for curing processes. Its synthesis involves the alkylation of acetophenone with ethyl iodide in the presence of a base, leading to the formation of the diethoxy derivative. The presence of the two ethoxy groups significantly enhances its ability to absorb UV light and generate free radicals, crucial for initiating polymerization. In application, 2,2-diethoxyacetophenone is predominantly used in the field of UV-curable coatings and adhesives. When exposed to UV light, it undergoes homolytic cleavage to produce reactive radicals that initiate the polymerization of acrylates and methacrylates. This property is highly valued in the production of high-performance coatings and adhesives, where rapid curing and strong adhesion are required. One of the major applications of 2,2-diethoxyacetophenone is in the manufacturing of durable coatings for various surfaces, including metals, plastics, and glass. These coatings are used in automotive, aerospace, and consumer goods industries, where they provide protection against environmental damage and enhance the aesthetic appeal of products. The compound’s efficiency in UV-induced polymerization ensures that coatings cure quickly, which is essential for high-speed production lines. Additionally, 2,2-diethoxyacetophenone is employed in the development of high-resolution lithographic systems. Its role in photoresists for microfabrication processes highlights its significance in the electronics industry. The ability to form precise patterns on semiconductor wafers and other substrates is critical for the fabrication of integrated circuits and other electronic components. The compound’s stability and reactivity make it a valuable component in the field of UV curing technology. Its effectiveness in initiating polymerization reactions has led to its widespread use in various industrial and technological applications. References 2024. Synthesis of Novel High-Performance Adsorbent Based on Modified Polyurethane/Polysaccharides for Wastewater Treatment. Journal of Polymers and the Environment, 32(11). DOI: 10.1007/s10924-024-03249-x 2021. Modular Synthesis of α-Substituted Alkenyl Acetals by a Palladium-Catalyzed Suzuki Reaction of α-Haloalkenyl Acetals with Organoboranes. Synlett, 32(06). DOI: 10.1055/a-1322-3916 1978. Chemistry of the biradicals produced in the Norrish Type II reaction. Research on Chemical Intermediates, 1(3). DOI: 10.1007/bf03155998 |
| Market Analysis Reports |