Online Database of Chemicals from Around the World
| Beijing Kaida Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bjkaida.cn | |||
 | +86 (10) 6299-3416 +86 13811060439 | |||
 | +86 (10) 6299-3416 | |||
 | kaida@bjkaida.cn bjkaida@163.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Gou Hebei Chemicals | India | |||
|---|---|---|---|---|
 | gouhebeichems.store | |||
 | +86 15874272473 | |||
 | info@gouhebeichems.store | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Telly Chemicals Pvt | China | |||
|---|---|---|---|---|
 | tellychemicals.ecer.com | |||
 | +852 52919765 | |||
 | +852 52919765 | |||
 | tellychemicals@protonmail.com | |||
 | Skype Chat | |||
 | WhatsApp:+61 485956223 | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Amine |
|---|---|
| Name | (S)-Methylenedioxymethamphetamine |
| Synonyms | (S)-(+)-3,4-(Methylenedioxy)methamphetamine; (S)-MDMA; (S)-N,alpha-Dimethyl-1,3-benzodioxole-5-ethanamine |
| Molecular Structure | 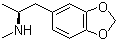 |
| Molecular Formula | C11H15NO2 |
| Molecular Weight | 193.24 |
| CAS Registry Number | 66142-89-0 |
| SMILES | C[C@@H](CC1=CC2=C(C=C1)OCO2)NC |
| Density | 1.1±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.536, Calc.* |
| Boiling Point | 283.4±9.0 °C (760 mmHg), Calc.* |
| Flash Point | 113.2±8.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
(S)-Methylenedioxymethamphetamine, commonly known as MDMA or "ecstasy," is a psychoactive substance known for its hallucinogenic and stimulant effects. MDMA was first synthesized in 1912 by chemists at Merck as a precursor for other drugs, but its psychoactive properties were not discovered until much later. The racemic mixture was first studied in the 1970s by chemist Alexander Shulgin, who noted its unique ability to enhance emotional experiences. The (S)-enantiomer C11H15NO2 is particularly well known for its potent psychoactive effects. Structurally, MDMA has a methylenedioxy ring attached to the phenylethylamine backbone, distinguishing it from other amphetamines by its additional oxygenated methylene bridge. MDMA works primarily by increasing the release of serotonin, dopamine, and norepinephrine in the brain. It binds to the transporters for these neurotransmitters, inhibiting their reuptake and causing a large increase in their extracellular concentrations. This mechanism underlies its euphoric and empathic effects, which improve mood, increase energy, emotional closeness, and alter sensory perception. MDMA gained popularity as a recreational drug in the late 20th century, particularly in the rave and dance club scenes. Users report feelings of euphoria, increased sociability, and heightened sensory perception, often accompanied by increased empathy and emotional connection. However, its use for recreational purposes carries risks such as dehydration, hyperthermia, and potential neurotoxicity, especially with repeated use or high doses. MDMA has recently been reexamined for its potential therapeutic benefits, particularly in the treatment of post-traumatic stress disorder (PTSD). Clinical trials have shown that MDMA-assisted psychotherapy can significantly reduce PTSD symptoms, providing patients with a controlled environment where MDMA's empathic effects help facilitate emotional processing and trauma resolution. The compound may help patients access and process difficult emotions with reduced anxiety, thereby increasing the effectiveness of psychotherapy. Due to its potential for abuse and neurotoxic effects, MDMA is classified as a Schedule I controlled substance in the United States and is similarly regulated internationally. This classification indicates that it has a high potential for abuse and no recognized medical uses. However, recent advances in clinical research have prompted a reconsideration of its legal status for therapeutic use under strict medical supervision. References 2017. The 2014 Philip S. Portoghese Medicinal Chemistry Lectureship: The “Phenylalkylaminome” with a Focus on Selected Drugs of Abuse. Journal of Medicinal Chemistry, 60(6). DOI: 10.1021/acs.jmedchem.7b00085 2014. Molecular basis of the selective binding of MDMA enantiomers to the alpha4beta2 nicotinic receptor subtype: Synthesis, pharmacological evaluation and mechanistic studies. European Journal of Medicinal Chemistry, 81. DOI: 10.1016/j.ejmech.2014.04.044 2002. Synthesis and capillary electrophoretic analysis of enantiomerically enriched reference standards of MDMA and its main metabolites. Bioorganic & Medicinal Chemistry, 10(4). DOI: 10.1016/s0968-0896(01)00367-4 |
| Market Analysis Reports |