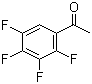
2',3',4',5'-Tetrafluoroacetophenone is an organic compound with the molecular formula C8H4F4O. It is a derivative of acetophenone, where four fluorine atoms are substituted at the para positions of the aromatic ring. The structure of the molecule features a carbonyl group attached to the aromatic ring, which is characteristic of acetophenones.
This chemical compound was first synthesized as part of research into the influence of fluorine substitution on the properties of aromatic compounds. The presence of fluorine atoms is known to significantly alter the physical and chemical properties of organic molecules, including their reactivity, solubility, and interaction with biological systems. Fluorine’s high electronegativity and small atomic size lead to increased stability of the molecule, making it an attractive candidate for various industrial and research applications.
2',3',4',5'-Tetrafluoroacetophenone has been used in organic synthesis as a versatile intermediate. The fluorine atoms on the aromatic ring increase the electrophilicity of the carbonyl group, making the compound more reactive in nucleophilic addition reactions. This property has made it a useful reagent for the synthesis of a variety of fluorinated organic compounds, which are valuable in the pharmaceutical and agrochemical industries.
In the pharmaceutical industry, the incorporation of fluorine atoms into organic molecules is a well-known strategy for enhancing metabolic stability, lipophilicity, and bioavailability. Fluorine substitution can also influence the binding affinity of compounds to biological targets, making fluorinated acetophenones like 2',3',4',5'-tetrafluoroacetophenone valuable as starting materials in drug development. Researchers have explored the use of such compounds in the design of molecules with enhanced pharmacological properties, especially in the context of cancer therapy, central nervous system disorders, and antimicrobial agents.
In materials science, 2',3',4',5'-tetrafluoroacetophenone and its derivatives can be used as building blocks for the synthesis of fluorinated polymers and materials. The introduction of fluorine atoms into polymer chains typically improves the chemical resistance, thermal stability, and surface properties of the materials. As a result, this compound has been investigated for potential use in the development of coatings, sensors, and other specialized materials that require specific performance characteristics.
Overall, 2',3',4',5'-tetrafluoroacetophenone is a fluorinated aromatic compound that serves as a key intermediate in organic synthesis. Its applications in the pharmaceutical and materials industries make it a valuable compound, particularly for the development of new drugs and advanced materials with enhanced properties. The combination of fluorine substitution and the acetophenone structure provides a foundation for a wide range of chemical reactions and product developments.
References
2012. Features of synthesis and structure of ethyl (2Z)-3-hydroxy-(2,3,4,5-tetrafluorophenyl)-propyl-2-enoate. Russian Journal of General Chemistry, 82(1).
DOI: 10.1134/s1070363212010197
|




























 GHS07 Warning Details
GHS07 Warning Details