Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Haichem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.haichem.com | |||
 | +86 (571) 8602-5531 | |||
 | +86 (571) 8602-6912 | |||
 | info@haichem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Ruijiang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ruicolor.com | |||
 | +86 (571) 2882-8110 | |||
 | +86 (571) 8885-4175 | |||
 | ruicolor01@vip.163.com orgchem001@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Yixing Xinyu Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xinyuchem.com | |||
 | +86 (510) 8744-6558 8744-1150 | |||
 | +86 (510) 8744-2808 / 8744-2345 | |||
 | sales@xinyuchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1993 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Chuan Shuo Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.transol.cn | |||
 | +86 (571) 8760-9021 | |||
 | qian@transol.cn | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2025 | ||||
| W & J PharmaChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.wjpharmachem.com | |||
 | +1 (301) 880-0624 | |||
 | +1 (301) 434-9366 | |||
 | info@wjpharmachem.com | |||
| Chemical manufacturer | ||||
| Xian Medihealth Company Ltd. | China | |||
|---|---|---|---|---|
 | www.xapharma.com | |||
 | +86 (29) 8786-1030 | |||
 | +86 (29) 8814-1462 | |||
 | xianmedihealth@gmail.com | |||
| Chemical distributor since 1992 | ||||
| Shanghai Shenqiang Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shenqiang.com | |||
 | +86 (21) 6297-1188 6297-1788 | |||
 | +86 (21) 6257-8811 | |||
 | shenqiang@shenqiang.com | |||
| Chemical manufacturer since 1996 | ||||
| Classification | Organic raw materials >> Amino compound >> Acyclic monoamines, polyamines and their derivatives and salts |
|---|---|
| Name | 2-(2,4-Diaminophenoxy)ethanol dihydrochloride |
| Molecular Structure | 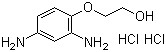 |
| Molecular Formula | C8H12N2O2.2(HCl) |
| Molecular Weight | 241.12 |
| CAS Registry Number | 66422-95-5 |
| EC Number | 266-357-1 |
| SMILES | C1=CC(=C(C=C1N)N)OCCO.Cl.Cl |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H319-H411 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P273-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P333+P317-P337+P317-P362+P364-P391-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||
|
2-(2,4-Diaminophenoxy)ethanol dihydrochloride is a chemical compound characterized by a phenoxyethanol backbone substituted with amino groups at the 2- and 4-positions of the aromatic ring, existing as a dihydrochloride salt. Its molecular structure consists of a 2,4-diaminophenyl group linked via an ether bond to an ethanol moiety, with the compound stabilized by two hydrochloride ions. The synthesis of 2-(2,4-diaminophenoxy)ethanol dihydrochloride generally involves the nucleophilic substitution of 2,4-dinitrophenol or its derivatives with ethylene oxide or 2-chloroethanol, followed by reduction of the nitro groups to amines. The formation of the dihydrochloride salt is achieved by treatment with hydrochloric acid, which protonates the amino groups, improving the compound’s solubility and stability. The presence of the dihydrochloride salt form makes the compound more amenable for storage and handling, reducing volatility and enhancing water solubility. This compound finds application primarily in pharmaceutical and biochemical research, often serving as an intermediate or building block in the synthesis of bioactive molecules. The 2,4-diaminophenyl moiety provides sites for further chemical modification, and the presence of the hydroxyethyl group allows for enhanced solubility and reactivity. Such structural features make it useful for designing compounds targeting enzyme active sites or receptor binding domains that interact with amine and hydroxyl functional groups. In addition to its synthetic utility, 2-(2,4-diaminophenoxy)ethanol dihydrochloride is employed in medicinal chemistry for the development of agents with antimicrobial, anticancer, or enzyme inhibitory properties. Its amino substitutions enable interactions through hydrogen bonding and ionic interactions, important for biological activity. The compound’s compatibility with aqueous environments, due to its salt form, facilitates its incorporation into various biological assay systems. Analytical techniques for characterization include nuclear magnetic resonance (NMR) spectroscopy, which confirms the substitution pattern and connectivity of the aromatic ring and side chain. Infrared (IR) spectroscopy identifies functional groups such as amino and hydroxyl, while mass spectrometry (MS) determines molecular weight and fragmentation profiles. Chromatographic methods like high-performance liquid chromatography (HPLC) are used to assess purity and monitor synthesis processes. Regarding safety, 2-(2,4-diaminophenoxy)ethanol dihydrochloride should be handled with standard laboratory precautions due to the potential irritant effects of aromatic amines and their salts. Protective gloves, eye protection, and working in well-ventilated areas or fume hoods are recommended to minimize exposure risks. There is no widespread evidence of severe toxicity, but care is taken to avoid unnecessary contact or inhalation. In summary, 2-(2,4-diaminophenoxy)ethanol dihydrochloride is a dihydrochloride salt of a hydroxyethyl-substituted diaminophenyl ether compound used mainly as a synthetic intermediate in pharmaceutical and biochemical research. Its chemical structure offers functional versatility for the development of biologically active molecules. Established synthetic routes, analytical characterization methods, and safety practices support its use in research and development contexts. References 1982. Mutagenic studies on the hair dye 2-(2',4'-diaminophenoxy) ethanol (DAPE) with different genetic systems. Mutation Research/Genetic Toxicology, 102(4). DOI: 10.1016/0165-1218(82)90137-9 1982. Lack of genotoxic activity of 2-(2',4'-diaminophenoxy)ethanol in Escherichia coli strains WP2, WP2uvrA and WP2uvrA/recA. Mutation Research/Genetic Toxicology, 102(4). DOI: 10.1016/0165-1218(82)90135-5 1982. Test for induction of chromosomal aberrations in Chinese hamster ovary cells (in vitro) by 2-(2',4'-diaminophenoxy)ethanol (DAPE). Mutation Research/Genetic Toxicology, 102(4). DOI: 10.1016/0165-1218(82)90139-2 |
| Market Analysis Reports |