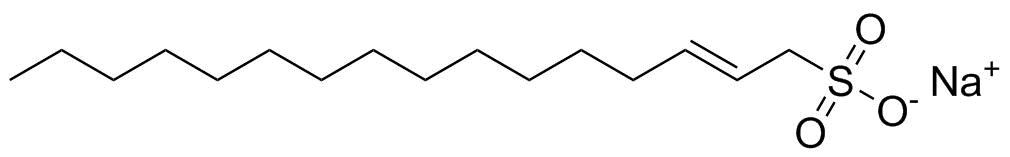
Sodium C14-16 olefin sulfonate is a synthetic anionic surfactant commonly used in personal care products, household cleaners, and industrial detergents. It is a mixture of sodium salts of sulfonated long-chain α-olefins, typically containing hydrocarbon chains with 14 to 16 carbon atoms. Its general structure consists of a hydrophobic alkyl chain attached to a hydrophilic sulfonate group (–SO3− Na+), giving it strong surface-active properties.
The compound is produced from linear α-olefins, which are hydrocarbons containing a terminal double bond. These olefins are derived from petrochemical feedstocks through processes such as ethylene oligomerization. The C14–C16 fraction is selected based on desired surfactant performance characteristics such as foaming, detergency, and solubility. The olefins are then subjected to sulfonation, typically using sulfur trioxide or related sulfonating agents, followed by neutralization with sodium hydroxide to form the sodium sulfonate salt.
The resulting product is not a single pure compound but rather a mixture of positional isomers and homologues, since sulfonation can occur at different positions along the carbon chain and across a range of chain lengths within the C14–C16 distribution. This structural diversity is typical of industrial surfactants and contributes to their broad functional performance.
Sodium C14-16 olefin sulfonate belongs to the class of anionic surfactants. In aqueous solution, it dissociates into sodium ions (Na+) and olefin sulfonate anions. The long hydrophobic hydrocarbon chain interacts with oils and nonpolar substances, while the sulfonate head group interacts strongly with water. This amphiphilic structure enables the formation of micelles above the critical micelle concentration, which is essential for its cleaning and emulsifying action.
The compound is widely used in shampoos, body washes, facial cleansers, and household cleaning products due to its strong detergency and foaming ability. It is often considered a high-performance cleansing agent capable of removing oils and particulate matter effectively, even in hard water conditions. Compared with some traditional sulfate surfactants, it can provide strong foam and cleaning performance while being formulated in blends to adjust mildness and viscosity.
In formulation systems, sodium C14-16 olefin sulfonate is frequently combined with amphoteric or nonionic surfactants to balance its strong cleansing power with improved skin compatibility. It contributes to foam richness and stability, which is an important sensory property in consumer cleaning products.
The physicochemical properties of sodium C14-16 olefin sulfonate are influenced by its hydrocarbon chain length distribution and the position of the sulfonate group. The long alkyl chains provide strong hydrophobic interactions, while the sulfonate group imparts high water solubility and ionic character. This combination results in efficient surface tension reduction and good emulsification of oily soils.
From a historical perspective, olefin sulfonates were developed as part of efforts to create high-performance synthetic detergents that could compete with or improve upon earlier surfactants such as alkyl sulfates and linear alkylbenzene sulfonates. Advances in petrochemical olefin production made it possible to tailor chain lengths and optimize surfactant properties for specific applications.
Overall, sodium C14-16 olefin sulfonate is a mixture of sodium salts of long-chain olefin sulfonic acids. Its strong anionic surfactant character, derived from a hydrophobic C14–C16 hydrocarbon backbone and a hydrophilic sulfonate group, makes it a widely used ingredient in modern cleaning and personal care formulations due to its effective cleansing, foaming, and emulsifying properties.
References
none
|