Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Taizhou Hengfeng Pharmaceutical & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hfchem.cn | |||
 | +86 (576) 8599-3534 | |||
 | +86 (576) 8599-3006 | |||
 | hfchem@hfchem.cn | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Biocaxis Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.biocaxis.com | |||
 | +86 (21) 6425-0676 | |||
 | +86 (21) 6425-0676 | |||
 | ribonucleotide@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Frapp's Chemical (NFTZ) Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.frappschem.com | |||
 | +86 (576) 8169-6106 | |||
 | +86 (576) 8169-6105 | |||
 | sales@frappschem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Win-Win Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.win-winchemical.com | |||
 | +86 (577) 6449-8589 +86 15325081899 | |||
 | +86 (577) 5699-4596 | |||
 | sales@win-winchemical.com winwinchemical@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Beckmann-Kenko GmbH | Germany | |||
|---|---|---|---|---|
 | www.beckmann-kenko.com | |||
 | +49 (4241) 930-888 | |||
 | +49 (4241) 930-889 | |||
 | info@beckmann-kenko.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Anax Laboratories | India | |||
|---|---|---|---|---|
 | www.anaxlab.com | |||
 | +91 (40) 6464-2010 | |||
 | info@anaxlab.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Berry & Associates, Inc. | USA | |||
|---|---|---|---|---|
 | www.berryassoc.com | |||
 | +1 (734) 426-3787 | |||
 | +1 (734) 426-9077 | |||
 | orders@berryassoc.com | |||
| Chemical manufacturer | ||||
| Molekula Ltd | UK | |||
|---|---|---|---|---|
 | www.molekula.com | |||
 | +44 (1747) 831-066 | |||
 | +44 (1747) 831-199 | |||
 | info@molekula.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Toronto Research Chemicals Inc. | Canada | |||
|---|---|---|---|---|
 | www.trc-canada.com | |||
 | +1 (416) 665-9696 | |||
 | +1 (416) 665-4439 | |||
 | info@trc-canada.com | |||
| Chemical manufacturer since 1982 | ||||
| Shanghai Desano Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.desano.com | |||
 | +86 (21) 5132-3300 | |||
 | +86 (21) 5132-3311 | |||
 | info@desano.com | |||
| Chemical manufacturer since 1996 | ||||
| Kemprotec Limited | UK | |||
|---|---|---|---|---|
 | www.kemprotec.com | |||
 | +44 (1642) 591-764 | |||
 | +44 (1845) 550-001 | |||
 | sales@kemprotec.co.uk | |||
| Chemical distributor since 1998 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Antiviral drugs |
|---|---|
| Name | Dideoxyinosine |
| Synonyms | 2',3'-Dideoxyinosine |
| Molecular Structure | 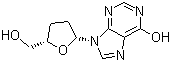 |
| Molecular Formula | C10H12N4O3 |
| Molecular Weight | 236.23 |
| Protein Sequence | N |
| CAS Registry Number | 69655-05-6 |
| EC Number | 614-994-4 |
| SMILES | C1C[C@@H](O[C@@H]1CO)N2C=NC3=C2N=CNC3=O |
| Solubility | 48 mg/mL (DMSO), <1 mg/mL (Water) (Expl.) |
|---|---|
| Density | 1.8±0.1 g/cm3, Calc.* |
| Melting point | 193 °C (Expl.) |
| Index of Refraction | 1.798, Calc.* |
| Boiling Point | 644.5±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 343.6±28.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335-H341-H372 Details | ||||||||||||
| Safety Statements | P203-P260-P261-P264-P264+P265-P270-P271-P280-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Dideoxyinosine (DDI), also known as 2',3'-dideoxyinosine, is a purine nucleoside analog used primarily in the treatment of human immunodeficiency virus (HIV) infections. It is an important member of a class of antiretroviral drugs known as nucleoside reverse transcriptase inhibitors (NRTIs). The discovery and development of DDI provided a significant advancement in the management of HIV/AIDS, especially during the early days of the epidemic. DDI was first synthesized in the 1980s as part of efforts to develop compounds that could inhibit the action of reverse transcriptase, an enzyme that is crucial for the replication of HIV. The structure of DDI closely resembles that of deoxyinosine, a natural nucleoside, but with a key modification: the absence of a 3'-hydroxy group on the sugar ring. This alteration prevents the enzyme from adding additional nucleotides during viral replication, effectively halting the process. The mechanism of action is similar to that of other NRTIs, which include drugs like zidovudine (AZT) and lamivudine (3TC). DDI was granted approval by the U.S. Food and Drug Administration (FDA) in 1991 for use in HIV treatment. It was initially marketed under the brand name Videx and was available as both oral capsules and powder for oral suspension. DDI works by incorporating into the viral DNA chain during replication, causing chain termination due to the lack of a 3'-hydroxy group that is necessary for further elongation of the DNA strand. In terms of clinical application, DDI was used as part of combination antiretroviral therapy (cART), a standard approach in HIV treatment that involves using multiple drugs with different mechanisms of action to reduce the risk of viral resistance. By inhibiting reverse transcriptase, DDI helps to lower the viral load in patients and improve immune system function, as measured by CD4 cell count. Although effective, DDI is associated with several side effects, including gastrointestinal issues, peripheral neuropathy, and pancreatitis. The drug was eventually superseded by other NRTIs that are better tolerated and have fewer side effects. However, it remains a key part of HIV treatment history and continues to be studied for its effects in various treatment regimens. DDI's discovery and application represent a milestone in the fight against HIV/AIDS. The drug demonstrated the potential of nucleoside analogs in treating viral infections, paving the way for the development of additional antiretroviral therapies that have significantly improved the prognosis of HIV-infected individuals. References 1990. Phase I Study of 2′,3′-Dideoxyinosine: Experience with 19 Patients at New York University Medical Center. Reviews of Infectious Diseases, 12(Suppl 5). DOI: 10.1093/clinids/12.supplement_5.s534 1994. Comparison of mitochondrial morphology, mitochondrial DNA content, and cell viability in cultured cells treated with three anti-human immunodeficiency virus dideoxynucleosides. Antimicrobial Agents and Chemotherapy, 38(8). DOI: 10.1128/aac.38.8.1824 2023. Long-term follow-up with multimodal imaging and functional testing in didanosine retinal toxicity. Journal Français d'Ophtalmologie, 46(6). DOI: 10.1016/j.jfo.2022.12.018 |
| Market Analysis Reports |