Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Chemical reagent >> Organic reagent >> Guanidine salt |
|---|---|
| Name | 1,1,3,3-Tetramethyl-2-(3-trimethoxysilylpropyl)guanidine |
| Synonyms | N-[Bis(dimethylamino)methylene]-3-(trimethoxysilyl)propylamine; N''-[3-(Trimethoxysilyl)propyl]tetramethylguanidine |
| Molecular Structure | 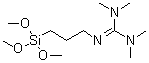 |
| Molecular Formula | C11H27N3O3Si |
| Molecular Weight | 277.44 |
| CAS Registry Number | 69709-01-9 |
| EC Number | 274-092-8 |
| SMILES | CN(C)C(=NCCC[Si](OC)(OC)OC)N(C)C |
| Solubility | Soluble (36 g/L) (25 °C), Calc.* |
|---|---|
| Density | 0.96±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.441, Calc.* |
| Boiling Point | 323.3±21.0 °C (760 mmHg), Calc.* |
| Flash Point | 149.4±22.1 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| Hazard Symbols |  GHS05 Danger Details GHS05 Danger Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H314-H318 Details | ||||||||||||
| Safety Statements | P260-P264-P264+P265-P280-P301+P330+P331-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P363-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
1,1,3,3-Tetramethyl-2-(3-trimethoxysilylpropyl)guanidine is a versatile chemical compound with significant applications in various fields, including materials science, catalysis, and surface modification. This compound consists of a guanidine core structure with trimethoxysilyl and tetramethyl groups, which contribute to its unique properties. The combination of these groups provides both high reactivity and the ability to interact with a range of surfaces, making it valuable in diverse industrial and research settings. The discovery of 1,1,3,3-tetramethyl-2-(3-trimethoxysilylpropyl)guanidine emerged from ongoing research into the development of functional silane compounds. Silanes have long been recognized for their ability to form strong bonds with various surfaces, particularly inorganic materials like glass, metals, and ceramics. The addition of the guanidine group to the structure enhanced the compound's reactivity, particularly in catalysis and surface modification processes. Guanidine-based compounds are known for their basicity and ability to interact with various organic and inorganic substrates, and when combined with silane functionality, they offer unique advantages in a range of applications. In terms of application, 1,1,3,3-tetramethyl-2-(3-trimethoxysilylpropyl)guanidine has found use in surface modification, particularly in the production of highly adhesive coatings and materials. The trimethoxysilyl group allows the compound to form strong covalent bonds with siliceous surfaces, improving adhesion between the surface and other materials. This property is particularly useful in the manufacturing of composites, coatings, and adhesives, where strong bonding to glass, metal, and ceramic surfaces is essential. The guanidine group provides additional functionality, allowing the compound to interact with other reactive groups or act as a catalyst in certain reactions. Another major application of 1,1,3,3-tetramethyl-2-(3-trimethoxysilylpropyl)guanidine lies in catalysis, particularly in organic reactions. The guanidine group imparts basicity to the compound, enabling it to act as a catalyst in various organic transformations. For example, it can be used as a base in the synthesis of functionalized organic compounds or in the activation of certain substrates. The presence of the trimethoxysilyl group also contributes to the compound's stability and solubility, making it useful in both homogeneous and heterogeneous catalytic systems. Additionally, 1,1,3,3-tetramethyl-2-(3-trimethoxysilylpropyl)guanidine has been explored for use in the development of advanced materials, particularly in the creation of hybrid organic-inorganic systems. The silane functionality allows the compound to integrate with inorganic materials, such as silica or metal oxides, to form materials with improved properties, such as increased mechanical strength, durability, or resistance to environmental degradation. These hybrid materials have applications in fields such as electronics, optics, and energy storage. Furthermore, the compound has potential applications in the modification of polymeric materials. Its ability to bond to polymer chains and impart additional functionality has made it of interest in the development of high-performance polymer composites. The presence of the guanidine group enhances the reactivity of the material, making it suitable for incorporation into advanced coatings, adhesives, and sealants, where strong bonding and resistance to environmental factors are critical. In summary, 1,1,3,3-tetramethyl-2-(3-trimethoxysilylpropyl)guanidine is a multifunctional compound with diverse applications across several industries. Its ability to modify surfaces, catalyze reactions, and form advanced hybrid materials makes it a valuable tool in the development of new materials and technologies. As research continues to explore its full potential, this compound is likely to see further use in areas ranging from coatings and adhesives to catalysis and polymer modification. References 2017. Graphene oxide covalently functionalized with an organic superbase as highly efficient and durable nanocatalyst for green Michael addition reaction. Research on Chemical Intermediates, 43(12). DOI: 10.1007/s11164-017-3105-2 2008. Aldol-Type Coupling of Ethyl Diazoacetate with SiO2-TMG. Synfacts, 2008(9). DOI: 10.1055/s-2008-1078660 |
| Market Analysis Reports |