Online Database of Chemicals from Around the World
| Unichem Company Ltd. | China | |||
|---|---|---|---|---|
 | www.unionchems.com | |||
 | +86 (510) 823-28705 | |||
 | webmaster@unionchems.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Zhanhua Darong Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.darongchem.com | |||
 | +86 (532) 6777-3200 +86 15689126900 | |||
 | +86 (532) 6777-3200 | |||
 | sales@darongchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Alfa Chemistry | USA | |||
|---|---|---|---|---|
 | www.alfa-chemistry.com | |||
 | +1 (201) 478-8534 | |||
 | +1 (516) 927-0118 | |||
 | inquiry@alfa-chemistry.com | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Xiangyang Runtongda Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.furundachemical.com | |||
 | +86 (710) 353-5121 +86 13508666631 | |||
 | +86 (710) 351-3703 | |||
 | xfurunda@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Synchemer Co., Limited | China | |||
|---|---|---|---|---|
 | www.synchemer.com | |||
 | +86 (20) 3668-5350 +86 13556109416 | |||
 | +86 (20) 3668-6430 | |||
 | info@synchemer.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Qingdao Riverside New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rivmat.com | |||
 | +86 (532) 8093-2738 +86 15689126900 | |||
 | +86 (532) 8093-2738 | |||
 | info@rivmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Eustar Functional Material Technology, Inc. | China | |||
|---|---|---|---|---|
 | www.eustar.com.cn | |||
 | +86 (0512) 5510-5882 | |||
 | purchaser@eustar.com.cn | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Catalysts and additives >> UV absorber |
|---|---|
| Name | Dimethyl 4-methoxybenzylidenemalonate |
| Synonyms | Dimethyl (p-methoxybenzylidene)malonate; Hostavin BRE 25; Hostavin PR 25; NSC 306435; PR 25; Sanduvor PR 25; Cyasorb UV 1988 |
| Molecular Structure | 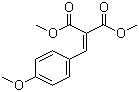 |
| Molecular Formula | C13H14O5 |
| Molecular Weight | 250.25 |
| CAS Registry Number | 7443-25-6 |
| EC Number | 231-185-8 |
| SMILES | COC1=CC=C(C=C1)C=C(C(=O)OC)C(=O)OC |
| Density | 1.2±0.1 g/cm3, Calc.*, 1.187 g/mL (Expl.) |
|---|---|
| Melting point | 56-57 °C (Expl.) |
| Index of Refraction | 1.540, Calc.* |
| Boiling Point | 335.5±27.0 °C (760 mmHg), Calc.* |
| Flash Point | 145.9±23.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H319-H411 Details | ||||||||||||
| Safety Statements | P264+P265-P273-P280-P305+P351+P338-P337+P317-P391-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Dimethyl 4-methoxybenzylidenemalonate is an organic compound widely recognized for its utility in organic synthesis, particularly as an intermediate in the production of pharmaceuticals and fine chemicals. This molecule, characterized by its benzylidene core and ester functionalities, plays a crucial role in creating diverse chemical frameworks due to its reactivity and structural versatility. Its discovery and use reflect the broader evolution of malonate derivatives in synthetic organic chemistry. The origins of dimethyl 4-methoxybenzylidenemalonate can be traced to research into malonic acid derivatives, which emerged as pivotal reagents in the mid-20th century for their ability to form carbon-carbon bonds. The introduction of the benzylidene group, combined with the electron-donating methoxy functionality, enhanced its applicability in specific transformations, including Michael additions, Knoevenagel condensations, and other base-catalyzed reactions. These features made the compound particularly attractive in synthetic pathways requiring selective reactivity. One of the primary applications of dimethyl 4-methoxybenzylidenemalonate is in the pharmaceutical industry. It serves as an intermediate in the synthesis of bioactive molecules, including heterocycles and aromatic derivatives. For instance, its utility in creating pyridine or coumarin scaffolds has been documented in the development of therapeutic agents with antimicrobial, anti-inflammatory, or anticancer properties. Additionally, the benzylidene malonate structure lends itself to fine-tuning reaction conditions, allowing chemists to introduce functional groups selectively during synthesis. In agrochemical applications, dimethyl 4-methoxybenzylidenemalonate is employed in the design of compounds with herbicidal and pesticidal properties. Its ability to form conjugated systems enables its incorporation into structures that interfere with biological pathways in target organisms. The compound's ease of derivatization also makes it suitable for the development of novel formulations aimed at improving efficacy and environmental safety. Beyond its role as an intermediate, dimethyl 4-methoxybenzylidenemalonate has also attracted attention in materials science. Its conjugated structure offers potential in the synthesis of organic semiconductors and light-emitting materials, where electron-rich aromatic systems are valuable. Although these applications are still emerging, preliminary studies suggest a promising future in advanced materials. Despite its synthetic advantages, considerations regarding the environmental and safety aspects of dimethyl 4-methoxybenzylidenemalonate are necessary. Proper handling, storage, and disposal protocols are critical to mitigate risks associated with its reactivity and potential impact on ecosystems. Ongoing research aims to develop more sustainable methods for its production and application, aligning with global efforts toward green chemistry. Dimethyl 4-methoxybenzylidenemalonate exemplifies the impact of functionalized malonate derivatives in modern organic chemistry. Its combination of reactivity, stability, and versatility ensures its continued relevance in diverse scientific and industrial domains. References 2019. Synthesis and Structure of 4-Het(aryl)-3,5,5-trimethoxycarbonyl-2-pyrrolidones. Russian Journal of General Chemistry, 89(7). DOI: 10.1134/s1070363219070260 2013. One-pot synthesis of polysubstituted 2-piperidinones from aromatic aldehydes, nitromethane, ammonium acetate, and dialkyl malonates. Monatshefte für Chemie - Chemical Monthly, 144(3). DOI: 10.1007/s00706-012-0912-4 2012. Selection, synthesis, and anti-inflammatory evaluation of the arylidene malonate derivatives as TLR4 signaling inhibitors. Bioorganic & Medicinal Chemistry, 20(20). DOI: 10.1016/j.bmc.2012.08.022 |
| Market Analysis Reports |