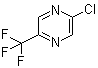
2-Chloro-5-(trifluoromethyl)pyrazine is an organic compound belonging to the pyrazine family, a class of heterocyclic aromatic compounds. This particular derivative is characterized by the presence of a chloro group at the 2-position and a trifluoromethyl group at the 5-position of the pyrazine ring.
The pyrazine structure itself consists of a six-membered ring containing two nitrogen atoms at non-adjacent positions, which contributes to its chemical reactivity and aromaticity. The halogen (chlorine) and trifluoromethyl (–CF₃) substituents on the ring significantly influence the compound's physical properties, such as its polarity, solubility, and reactivity.
The trifluoromethyl group (–CF₃) is a strong electron-withdrawing group, which can alter the electron density on the aromatic ring, often making the compound more reactive in electrophilic substitution reactions. This group is widely recognized for its influence in enhancing the lipophilicity and stability of compounds, as well as for improving the performance of certain chemical reactions. The chlorine atom at the 2-position also contributes to the compound’s electronic properties, potentially enhancing its reactivity in synthetic applications.
2-Chloro-5-(trifluoromethyl)pyrazine has various applications, particularly in the field of organic synthesis and materials science. The unique combination of functional groups on the pyrazine ring makes it useful as a building block in the synthesis of more complex molecules, including pharmaceuticals, agrochemicals, and other fine chemicals. The trifluoromethyl group is often incorporated into molecules to enhance their bioactivity, stability, or ability to interact with biological targets, making this compound of interest in drug discovery and development.
Furthermore, the compound's ability to engage in reactions such as nucleophilic substitution or condensation is important for the preparation of pyrazine derivatives, which are used in a wide range of applications, including in the manufacture of dyes, coatings, and other specialty chemicals. The electronic and steric properties of the substituents can be finely tuned for specific industrial applications, making compounds like 2-chloro-5-(trifluoromethyl)pyrazine valuable in diverse chemical processes.
While its direct biological or pharmacological applications may require further research, the structure and substituent effects of 2-chloro-5-(trifluoromethyl)pyrazine suggest its potential use in the design of bioactive molecules, particularly those that require specific interactions with receptors or enzymes. As such, it might be investigated further for its utility in medicinal chemistry, where the influence of fluorine and chlorine on compound behavior is well-documented.
In conclusion, 2-chloro-5-(trifluoromethyl)pyrazine is a versatile chemical with several potential applications in the synthesis of more complex organic molecules, particularly in the fields of pharmaceuticals, agrochemicals, and specialty materials. The presence of the trifluoromethyl and chloro groups provides it with unique properties that are valuable in both synthetic chemistry and materials science.
|












 GHS06 Danger Details
GHS06 Danger Details