Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphine ligand |
|---|---|
| Name | Bis(4-bromophenyl)phenylphosphine |
| Molecular Structure | 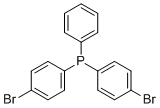 |
| Molecular Formula | C18H13Br2P |
| Molecular Weight | 420.08 |
| CAS Registry Number | 84591-80-0 |
| SMILES | C1=CC=C(C=C1)P(C2=CC=C(C=C2)Br)C3=CC=C(C=C3)Br |
| Boiling Point | 432.8±30.0 °C (760 mmHg), Calc.* |
|---|---|
| Flash Point | 215.6±24.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Damger Details GHS07;GHS08 Damger Details |
|---|---|
| Risk Statements | H302-H315-H319-H372-H373-H317-H335-H413 Details |
| Safety Statements | P501-P273-P272-P260-P270-P271-P264-P280-P302+P352-P314-P337+P313-P305+P351+P338-P362+P364-P333+P313-P301+P312+P330-P304+P340+P312-P403+P233-P405 Details |
| SDS | Available |
|
Bis(4-bromophenyl)phenylphosphine is a notable compound in the field of organophosphorus chemistry, recognized for its utility in catalysis and material science. This compound features a central phosphorus atom bonded to three phenyl groups, with two of these groups being 4-bromophenyl. The presence of bromine atoms on the phenyl rings enhances its reactivity and facilitates various applications in organic synthesis. The synthesis of Bis(4-bromophenyl)phenylphosphine typically involves the reaction of a suitable phenylphosphine precursor with bromobenzene. The introduction of bromine substituents on the phenyl rings increases the electron-withdrawing effect, which influences the electronic properties of the phosphorus center. This modification has been shown to impact the compound's reactivity and its interactions in catalytic processes. One significant application of Bis(4-bromophenyl)phenylphosphine is in the field of homogeneous catalysis. Its role as a ligand in palladium-catalyzed cross-coupling reactions, such as Suzuki and Heck reactions, has been extensively studied. The electron-withdrawing bromine atoms in the phenyl rings improve the stability and efficiency of the palladium complex, enhancing the overall catalytic performance. This improvement is particularly valuable in the synthesis of complex organic molecules where high reactivity and selectivity are required. In addition to its role in catalysis, Bis(4-bromophenyl)phenylphosphine has applications in material science. It is used in the preparation of advanced materials such as organic light-emitting diodes (OLEDs) and liquid crystals. The compound's ability to coordinate with metal centers makes it a valuable component in the design of new materials with tailored properties. For example, in OLED technology, the phosphine ligand can help in the formation of metal-organic frameworks that exhibit desirable optical properties. The compound's utility extends to the development of new methods for organic synthesis. Its unique electronic and steric properties make it a useful tool for exploring new reaction pathways and improving existing protocols. Researchers have utilized Bis(4-bromophenyl)phenylphosphine to develop more efficient synthesis methods for pharmaceuticals and agrochemicals. The ability to fine-tune its reactivity by altering the substituents on the phenyl rings allows for the optimization of reaction conditions and outcomes. Further research into Bis(4-bromophenyl)phenylphosphine has focused on understanding its interactions with various metal centers and its behavior in different chemical environments. Studies have explored its potential in catalyzing reactions involving heteroatoms and its performance in asymmetric synthesis. These investigations contribute to a deeper understanding of how modifications to the phosphine ligand can influence its catalytic properties and applications. Overall, Bis(4-bromophenyl)phenylphosphine represents an important advancement in organophosphorus chemistry. Its versatility and effectiveness in various applications make it a valuable tool for chemists working in both academic and industrial settings. As research continues, new applications and improvements are expected, further establishing its significance in the field. References 2020. Effects of Different Eelectron-withdrawing Moieties on the General Photoelectric Properties of Fluorene-based Dimers. Chemical Research in Chinese Universities, 36(4). DOI: 10.1007/s40242-020-0191-2 2012. Cross Coupling of Primary Phosphines. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-208-00216 |
| Market Analysis Reports |