Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Aryl compounds >> Biphenyl compounds |
|---|---|
| Name | 2-(Di-tert-butylphosphino)-3,4,5,6-tetramethyl-2',4',6'-triisopropyl-1,1'-biphenyl |
| Synonyms | Di-tert-butyl(2',4',6'-triisopropyl-3,4,5,6-tetramethylbiphenyl-2-yl)phosphine |
| Molecular Structure | 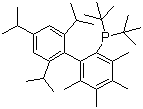 |
| Molecular Formula | C33H53P |
| Molecular Weight | 480.75 |
| CAS Registry Number | 857356-94-6 |
| EC Number | 813-582-3 |
| SMILES | CC1=C(C(=C(C(=C1C)C2=C(C=C(C=C2C(C)C)C(C)C)C(C)C)P(C(C)(C)C)C(C)(C)C)C)C |
| Melting point | 162-163 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335-H412 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P273-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P312-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
2-(Di-tert-butylphosphino)-3,4,5,6-tetramethyl-2',4',6'-triisopropyl-1,1'-biphenyl is a sophisticated ligand known for its role in enhancing the efficiency of metal-catalyzed reactions. This compound features a complex structure with a biphenyl core substituted with bulky groups: di-tert-butylphosphino and a combination of tetramethyl and triisopropyl groups. The unique combination of these substituents imparts significant steric and electronic properties to the ligand. The discovery of this ligand arose from the need to optimize catalytic processes by improving both the stability and selectivity of metal catalysts. The bulky di-tert-butylphosphino groups offer substantial steric hindrance, which helps to stabilize the metal center and prevent unwanted side reactions. The tetramethyl and triisopropyl substitutions further enhance the ligand's ability to shield the metal center while fine-tuning its electronic environment. This ligand is primarily employed in palladium-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura and Heck reactions. These reactions are essential in organic synthesis for forming carbon-carbon bonds, which are pivotal for the construction of complex organic molecules. By stabilizing the palladium catalyst and optimizing its electronic properties, this ligand contributes to higher reaction yields and improved control over selectivity. In addition to cross-coupling reactions, the ligand finds applications in other catalytic processes, including hydrogenation and carbonylation. Its ability to stabilize transition metal centers while modulating their electronic environment makes it a versatile tool for various catalytic transformations. The development of 2-(Di-tert-butylphosphino)-3,4,5,6-tetramethyl-2',4',6'-triisopropyl-1,1'-biphenyl exemplifies the advancements in ligand design aimed at improving catalytic performance. The strategic combination of steric and electronic effects in this ligand demonstrates its value in both academic research and industrial applications. Overall, this compound represents a significant contribution to the field of organometallic chemistry, showcasing how advanced ligand design can enhance the efficiency and selectivity of catalytic reactions. References 2022. Recent Developments in N2-Selective Functionalizations of 1,2,3-Triazoles. Synthesis, 54(06). DOI: 10.1055/s-0040-1719902 2022. How Rhodium(I)-Catalyzed Phosphorus(III)-Directed C-H Bond Functionalizations Can Improve the Catalytic Activities of Phosphines. Synlett, 33(01). DOI: 10.1055/s-0041-1737325 2007. Pd-Catalyzed Amidations of Aryl Chlorides Using Monodentate Biaryl Phosphine Ligands: A Kinetic, Computational, and Synthetic Investigation. Journal of the American Chemical Society, 129(42). DOI: 10.1021/ja0717414 |
| Market Analysis Reports |