Online Database of Chemicals from Around the World
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hangzhou Colorific Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.colorific-chem.com | |||
 | +86 (571) 8971-2978 | |||
 | +86 (571) 8971-2976 | |||
 | zhu_xuguang@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wuhan Silworld Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.silworldchemical.com | |||
 | +86 (27) 8561-3400 +86 15827173649 | |||
 | +86 (27) 8561-3419 | |||
 | info@silworldchemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Hongda Group | China | |||
|---|---|---|---|---|
 | www.hongda-chem.com | |||
 | +86 (574) 8790-7888 | |||
 | +86 (574) 8726-5831 | |||
 | chemcolor@hongda-chem.com | |||
| Chemical manufacturer since 1992 | ||||
| Anisyn, Inc. | USA | |||
|---|---|---|---|---|
 | www.anisyn.com | |||
 | +1 (269) 372-8736 | |||
 | +1 (269) 372-3397 | |||
 | sales@anisyn.com | |||
| Chemical manufacturer | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Norquay Technology Inc. | USA | |||
|---|---|---|---|---|
 | www.norquaytech.com | |||
 | +1 (610) 874-4330 | |||
 | +1 (610) 874-3575 | |||
 | sales@norquaytech.com | |||
| Chemical manufacturer since 1987 | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Anshan Beida Synthetic Chemicals Factory | China | |||
|---|---|---|---|---|
 | www.chinabeidachem.com | |||
 | +86 (412) 841-0999 843-9888 | |||
 | +86 (412) 841-5555 / 843-9333 | |||
 | beida@chinabeidachem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Quinoline compound |
|---|---|
| Name | Carbazole |
| Synonyms | 9-Azafluorene ; Dibenzopyrrole ; Diphenylenimine; Dibenzopyrrole; Diphenylenimine; 9-Azafluorene |
| Molecular Structure | 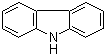 |
| Molecular Formula | C12H9N |
| Molecular Weight | 167.21 |
| CAS Registry Number | 86-74-8 |
| EC Number | 201-696-0 |
| SMILES | C1=CC=C2C(=C1)C3=CC=CC=C3N2 |
| Density | 1.1 |
|---|---|
| Melting point | 240-246 °C |
| Boiling point | 355 °C |
| Flash point | 220 °C |
| Water solubility | <0.1 g/100 mL at 19 °C |
| Hazard Symbols |   GHS08;GHS09 Warning Details GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H341-H351-H400-H411-H413 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P203-P273-P280-P318-P391-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Carbazole, an organic compound with the formula C12H9N, consists of a fused ring structure that combines a dibenzofuran and a nitrogen atom. Discovered in the 19th century, carbazole was first isolated from coal tar in 1883 by the German chemist Otto Dimroth. Since its initial discovery, carbazole has garnered significant attention for its unique chemical properties and potential applications across various fields. The structure of carbazole is characterized by a central nitrogen atom located within a system of fused benzene rings. This arrangement provides carbazole with aromatic characteristics, contributing to its stability and reactivity. Over the years, carbazole has been synthesized through various methods, including the cyclization of phenyl hydrazine and other nitrogen-containing compounds, as well as via the oxidation of carbazole derivatives. Its synthesis and modification have opened pathways for diverse applications. Carbazole's primary applications lie in the fields of organic electronics and materials science. One of its most notable uses is as a building block in the manufacture of organic light-emitting diodes (OLEDs). The excellent electron-transporting properties of carbazole make it an ideal candidate for the fabrication of OLED materials, which are pivotal in modern display technologies. Carbazole-based compounds are often utilized as host materials or dopants in OLEDs, contributing to efficient light emission and improved device performance. In addition to its role in OLEDs, carbazole is employed in the production of various polymers and resins. Its incorporation into polymer matrices enhances thermal stability and mechanical properties, making carbazole-containing materials suitable for applications in coatings, adhesives, and engineering plastics. Furthermore, carbazole derivatives are utilized as fluorescent dyes and pigments in various consumer products, including plastics and inks. The pharmaceutical industry has also recognized the significance of carbazole due to its biological activities. Numerous carbazole derivatives have demonstrated promising pharmacological properties, including antimicrobial, anti-inflammatory, and anticancer activities. Research is ongoing to explore the therapeutic potential of these derivatives, leading to the development of new drugs and treatments. Despite its numerous applications, the environmental impact of carbazole and its derivatives has raised concerns. As a byproduct of coal tar and petroleum refining, carbazole can be released into the environment, leading to potential toxicity in aquatic organisms. Consequently, there is an increasing emphasis on developing environmentally friendly synthesis methods and exploring sustainable alternatives in the production of carbazole and its derivatives. In conclusion, carbazole is a versatile organic compound with a rich history and wide-ranging applications. From its discovery in coal tar to its critical role in modern electronic devices and materials, carbazole continues to be a focal point of research and development across various scientific disciplines. As innovations in synthesis and applications evolve, carbazole is poised to play an increasingly important role in the advancement of technology and materials science. References 2015. Synthesis, in vitro antimicrobial and cytotoxic activities of new carbazole derivatives of ursolic acid. Bioorganic & Medicinal Chemistry Letters, 25(3). DOI: 10.1016/j.bmcl.2014.12.021 2013. Investigation of carbazole degradation by the sono-Fenton process. Water science and technology : a journal of the International Association on Water Pollution Research, 68(7). DOI: 10.2166/wst.2013.245 2012. Carbazole-thiosemicarbazone-Hg(ii) ensemble-based colorimetric and fluorescence turn-on toward iodide in aqueous media and its application in live cell imaging. Organic & Biomolecular Chemistry, 10(16). DOI: 10.1039/c2ob06792g |
| Market Analysis Reports |