Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Fluoropharm Co.,Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoropharm.com | |||
 | +86 (571) 8558-6753 +86 13336034509 | |||
 | +86 (571) 8558-6753 | |||
 | sales@fluoropharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Fevipiprant |
| Synonyms | QAW 039; [1-(4-((Methane)sulfonyl)-2-trifluoromethylbenzyl)-2-methyl-1H-pyrrolo[2,3-b]pyridin-3-yl]acetic acid |
| Molecular Structure | 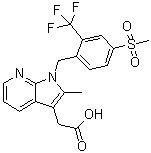 |
| Molecular Formula | C19H17F3N2O4S |
| Molecular Weight | 426.41 |
| CAS Registry Number | 872365-14-5 |
| SMILES | CC1=C(C2=C(N1CC3=C(C=C(C=C3)S(=O)(=O)C)C(F)(F)F)N=CC=C2)CC(=O)O |
| Solubility | Practically insoluble (0.043 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.44±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.597, Calc.* |
| Boiling Point | 637.6±55.0 °C (760 mmHg), Calc.* |
| Flash Point | 339.4±31.5 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2016 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
Fevipiprant, also known by its development code QAW039, is a potent and selective antagonist of the prostaglandin D2 receptor 2 (DP2), also known as the chemoattractant receptor-homologous molecule expressed on Th2 cells (CRTh2). This compound was developed to target inflammatory pathways associated with allergic and eosinophilic conditions, particularly asthma and other respiratory diseases. Its discovery marked a significant step forward in the pursuit of personalized treatments for inflammatory disorders. The development of Fevipiprant emerged from extensive research into the role of prostaglandin D2 (PGD2) in the immune system. PGD2 is a lipid mediator that plays a key role in allergic inflammation, acting through its DP1 and DP2 receptors. The DP2 receptor is expressed on Th2 lymphocytes, eosinophils, and basophils, making it a critical target for conditions driven by Th2 inflammation. By selectively inhibiting DP2, Fevipiprant blocks the recruitment and activation of inflammatory cells, offering a novel mechanism to modulate the immune response in diseases like asthma. Preclinical studies of Fevipiprant demonstrated its ability to inhibit DP2-mediated cellular responses, including the migration of eosinophils and Th2 cells to inflammatory sites. These findings laid the groundwork for its clinical evaluation as a therapeutic agent. Fevipiprant advanced into clinical trials as a promising candidate for the treatment of moderate-to-severe asthma, where current therapies such as inhaled corticosteroids and β2-agonists often fall short for patients with uncontrolled symptoms. Clinical trials revealed that Fevipiprant significantly reduced airway inflammation, as evidenced by lower sputum eosinophil counts, a biomarker associated with severe asthma exacerbations. The drug also showed potential to improve lung function and reduce exacerbations in patients with eosinophilic asthma, a phenotype characterized by high levels of eosinophilic inflammation. Its oral administration provided a convenient alternative to injectable biologics targeting similar pathways, such as anti-IL-5 therapies. Beyond asthma, Fevipiprant has been investigated for potential applications in other inflammatory diseases, including chronic rhinosinusitis with nasal polyps and atopic dermatitis. The broad role of PGD2 and DP2 in allergic inflammation makes Fevipiprant a candidate for treating a range of conditions driven by similar immune mechanisms. Despite its promising early results, further clinical development of Fevipiprant faced challenges. Larger phase III trials failed to demonstrate significant improvements in asthma control compared to placebo, leading to the discontinuation of its development for asthma by Novartis, its primary developer. These outcomes highlighted the complexity of targeting specific immune pathways and underscored the need for a deeper understanding of patient-specific responses to such treatments. Fevipiprant’s journey represents the ongoing effort to develop targeted therapies for inflammatory diseases. Its selective inhibition of DP2 remains a valuable pharmacological insight, contributing to the broader exploration of lipid mediators and their receptors in drug discovery. While its clinical development in asthma has ceased, the knowledge gained from Fevipiprant’s studies continues to inform research into innovative treatments for immune-mediated disorders. References 2021. Streamlined Synthesis of Fevipiprant. Synfacts, 17(10). DOI: 10.1055/s-0040-1720917 2015. Synthesis of a high specific activity methyl sulfone tritium isotopologue of fevipiprant (NVP‐QAW039). Journal of Labelled Compounds and Radiopharmaceuticals, 58(4). DOI: 10.1002/jlcr.3281 2021. Long-term safety and exploratory efficacy of fevipiprant in patients with inadequately controlled asthma: the SPIRIT randomised clinical trial. Respiratory Research, 22(1). DOI: 10.1186/s12931-021-01904-8 |
| Market Analysis Reports |