Online Database of Chemicals from Around the World
| Zhejiang Yangfan New Materials Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shoufu.com | |||
 | +86 (571) 8890-2510 8890-2504 8890-2511 | |||
 | +86 (571) 8890-2500 | |||
 | shoufu@shoufuchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Chiron AS | Norway | |||
|---|---|---|---|---|
 | www.chiron.no | |||
 | +47 (73) 874-490 | |||
 | +47 (73) 874-499 | |||
 | chiron@chiron.no | |||
| Chemical manufacturer | ||||
| Sinova Corporation | USA | |||
|---|---|---|---|---|
 | www.sinovainc.com | |||
 | +1 (301) 961-1525 | |||
 | +1 (240) 235-4288 | |||
 | sales@sinovainc.com | |||
| Chemical manufacturer | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Thiol/thiophenol |
|---|---|
| Name | Diphenyl disulfide |
| Synonyms | Phenyl disulfide |
| Molecular Structure | 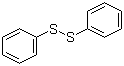 |
| Molecular Formula | C12H10S2 |
| Molecular Weight | 218.34 |
| CAS Registry Number | 882-33-7 |
| EC Number | 212-926-4 |
| FEMA | 3225 |
| SMILES | C1=CC=C(C=C1)SSC2=CC=CC=C2 |
| Melting point | 58-61 °C |
|---|---|
| Boiling point | 191-192 °C |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335-H400-H410 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P273-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| Transport Information | UN 3335 | ||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Diphenyl disulfide (DPDS) is an organic compound consisting of two benzene rings connected by a disulfide bond. It was discovered in the late 19th century during the study of sulfur-containing organic compounds. Its synthesis was first reported by Arthur Michael in 1889, using iodine to oxidize thiophenol. DPDS can also be synthesized by oxidizing thiophenol using hydrogen peroxide or other oxidants. Its chemical structure is two phenyl groups connected by a disulfide bond. It is a white crystalline solid that is soluble in organic solvents such as benzene, toluene, and chloroform. The disulfide bond in DPDS can be easily reduced to produce two thiophenol molecules, making it a useful oxidant and reductant. The presence of aromatic rings gives it unique electronic properties that affect its reactivity and interactions in various applications. DPDS is often used to introduce disulfide bonds in organic molecules, which is essential for the synthesis of various sulfur-containing compounds. It is also used in thiol-disulfide exchange reactions, which are important for studying protein folding and modifying biomolecules. It acts as a ligand in transition metal catalysis, helping to form composite catalysts that promote various chemical reactions, including cross-coupling and oxidation processes. DPDS is used as a stabilizer in polymers to prevent degradation by neutralizing free radicals, thereby improving the durability and service life of plastic materials. It is used as a vulcanizing agent in rubber production to help form sulfur crosslinks, which enhance the elasticity and strength of rubber products. DPDS is used in the synthesis of prodrugs, in which disulfide bonds are cleaved in vivo to release the active drug. Due to its ability to interact with thiol groups in proteins, it has been studied for its potential as an enzyme inhibitor and a candidate for therapeutic applications. DPDS is used to remove mercury from industrial wastewater, forming a stable complex that can be easily removed from the wastewater. It is used in processes to recover elemental sulfur from industrial wastewater, which helps in environmental protection and resource recovery. DPDS is used in redox titrations and studies to explore redox behavior and develop analytical methods to detect and quantify various analytes. In protein chemistry, DPDS modifies proteins by forming disulfide bonds, which helps in studying protein structure and function. DPDS should be handled with care. It can cause skin and eye irritation and should be stored in a cool, dry place away from light and heat sources. Personal protective equipment such as gloves and goggles should be used during handling. References 2023. Use of the Redox Properties of Hydrazine in the Synthesis of Organochalcogen Compounds (A Review). Russian Journal of General Chemistry, 93(14). DOI: 10.1134/s107036322314030x 2023. Organic Sulfur Derivatives and Their Metal Complexes as Promising Pharmacologically Active Compounds. Russian Journal of Coordination Chemistry, 49(12). DOI: 10.1134/s1070328423600894 2023. Modern Trends in the Synthesis of Disulfides: From Metal-Containing Catalysts to Nonmaterial Reagents (Review). Russian Journal of Coordination Chemistry, 49(12). DOI: 10.1134/s1070328423600985 |
| Market Analysis Reports |