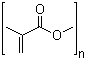
Methacrylic acid methyl ester polymers, commonly referred to as polymethyl methacrylate (PMMA), are synthetic polymers derived from the polymerization of methyl methacrylate (MMA) monomers. These polymers belong to the family of acrylic polymers and are characterized by their transparent, rigid, and weather-resistant properties. The polymer backbone consists of repeating units of methyl methacrylate with the general structure –[CH2–C(CH3)(COOCH3)]–n, where n represents the degree of polymerization.
The discovery of polymethyl methacrylate dates back to the early 20th century, with significant development during the 1930s. The polymerization of methyl methacrylate was achieved through free radical mechanisms, allowing for controlled synthesis and high molecular weight polymers. Advances in polymerization techniques such as bulk, solution, emulsion, and suspension polymerization have enabled the production of PMMA with tailored molecular weights and properties to meet diverse application requirements.
PMMA is widely used due to its optical clarity, resistance to ultraviolet light, and excellent weatherability. These properties have made it a popular alternative to glass in numerous applications, including lenses, automotive windows, skylights, and display screens. Its lightweight nature combined with high impact resistance enhances safety and ease of handling compared to traditional glass materials.
In addition to optical uses, methacrylic acid methyl ester polymers serve as key materials in coatings, adhesives, and sealants. Their ability to form durable films with good adhesion to various substrates makes them suitable for protective and decorative coatings in construction and automotive industries. Modified PMMA copolymers are also employed to improve flexibility, chemical resistance, or mechanical strength depending on the specific application.
The biomedical field utilizes PMMA for applications such as bone cement in orthopedic surgery and as a material for dental prosthetics. Its biocompatibility and ease of processing allow for the fabrication of custom implants and dental devices. Research continues into enhancing the polymer’s properties for improved biostability and mechanical performance in medical applications.
PMMA exhibits excellent electrical insulation properties and thermal stability up to moderate temperatures, contributing to its use in electronic and electrical components. The polymer’s resistance to solvents and chemicals varies with its formulation, but generally, it is stable against many common substances. Its relatively low glass transition temperature allows for ease of thermoforming into complex shapes.
Environmental considerations surrounding PMMA focus on its non-biodegradability and challenges in recycling. Efforts to improve sustainability have included developing chemical recycling methods and incorporating bio-based monomers to reduce dependence on fossil-derived raw materials. Additionally, research into copolymers and blends aims to enhance degradability while maintaining performance.
Overall, methacrylic acid methyl ester polymers have played a pivotal role in advancing materials science with their combination of optical clarity, mechanical robustness, and chemical resistance. Their extensive use across industries from automotive to healthcare underscores their versatility and ongoing relevance in modern polymer technology.
References
none
|



























 GHS07 Warning Details
GHS07 Warning Details