Online Database of Chemicals from Around the World
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2010 | ||||
| Classification | API >> Hormone and endocrine-regulating drugs >> Pancreatic hormones and other blood sugar regulating drugs |
|---|---|
| Name | Semaglutide |
| Synonyms | NN 9535; NNC 0113-0217;18-[[(1R)-4-[2-[2-[2-[2-[2-[2-[[(5S)-5-[[(2S)-2-[[(2S)-2-[[(2S)-5-amino-2-[[2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S)-2-[[(2S,3R)-2-[[(2S)-2-[[(2S,3R)-2-[[2-[[(2S)-2-[[2-[[(2S)-2-amino-3-(1H-imidazol-5-yl)propanoyl]amino]-2-methylpropanoyl]amino]-4-carboxybutanoyl]amino]acetyl]amino]-3-hydroxybutanoyl]amino]-3-phenylpropanoyl]amino]-3-hydroxybutanoyl]amino]-3-hydroxypropanoyl]amino]-3-carboxypropanoyl]amino]-3-methylbutanoyl]amino]-3-hydroxypropanoyl]amino]-3-hydroxypropanoyl]amino]-3-(4-hydroxyphenyl)propanoyl]amino]-4-methylpentanoyl]amino]-4-carboxybutanoyl]amino]acetyl]amino]-5-oxopentanoyl]amino]propanoyl]amino]propanoyl]amino]-6-[[(2S)-1-[[(2S)-1-[[(2S,3S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-5-carbamimidamido-1-[[2-[[(2S)-5-carbamimidamido-1-(carboxymethylamino)-1-oxopentan-2-yl]amino]-2-oxoethyl]amino]-1-oxopentan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-1-oxopropan-2-yl]amino]-3-methyl-1-oxopentan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-4-carboxy-1-oxobutan-2-yl]amino]-6-oxohexyl]amino]-2-oxoethoxy]ethoxy]ethylamino]-2-oxoethoxy]ethoxy]ethylamino]-1-carboxy-4-oxobutyl]amino]-18-oxooctadecanoic acid |
| Molecular Structure | 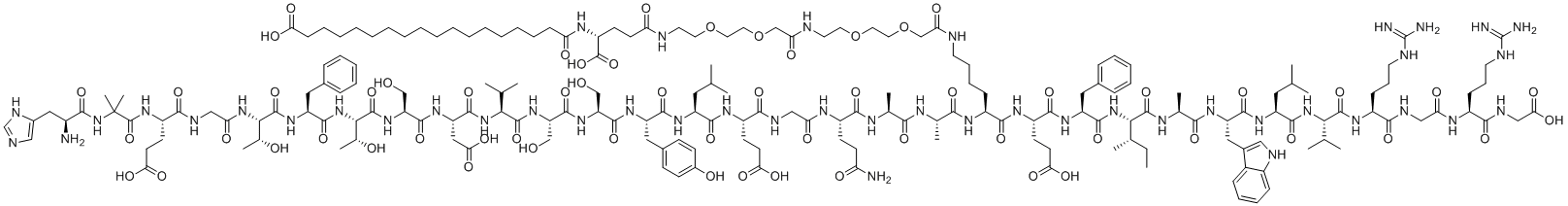 |
| Molecular Formula | C187H291N45O59 |
| Molecular Weight | 4113.58 |
| CAS Registry Number | 910463-68-2 |
| EC Number | 691-729-9 |
| SMILES | CC[C@H](C)[C@@H](C(=O)N[C@@H](C)C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCNC(=N)N)C(=O)NCC(=O)N[C@@H](CCCNC(=N)N)C(=O)NCC(=O)O)NC(=O)[C@H](CC3=CC=CC=C3)NC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CCCCNC(=O)COCCOCCNC(=O)COCCOCCNC(=O)CC[C@H](C(=O)O)NC(=O)CCCCCCCCCCCCCCCCC(=O)O)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(=O)N)NC(=O)CNC(=O)[C@H](CCC(=O)O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC4=CC=C(C=C4)O)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H](C(C)C)NC(=O)[C@H](CC(=O)O)NC(=O)[C@H](CO)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC5=CC=CC=C5)NC(=O)[C@H]([C@@H](C)O)NC(=O)CNC(=O)[C@H](CCC(=O)O)NC(=O)C(C)(C)NC(=O)[C@H](CC6=CN=CN6)N |
| Hazard Symbols |  GHS08 Warning Details GHS08 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, was developed by Novo Nordisk and gained FDA approval in 2017 for the treatment of type 2 diabetes. The discovery of semaglutide followed extensive research into the GLP-1 hormone, which is involved in regulating blood sugar levels. Researchers sought to create a more stable and longer-acting analog of GLP-1 to improve glycemic control and patient compliance. Semaglutide was engineered with modifications to extend its half-life, allowing for once-weekly dosing. Semaglutide is used to improve blood sugar control in adults with type 2 diabetes. By mimicking the action of the GLP-1 hormone, it stimulates insulin secretion, inhibits glucagon release, and slows gastric emptying, which helps regulate blood glucose levels. Its once-weekly injection regimen enhances patient compliance compared to daily medications. Semaglutide is also approved for chronic weight management under the brand name Wegovy. It helps reduce body weight by increasing feelings of fullness and decreasing appetite, leading to lower calorie intake. Semaglutide has shown cardiovascular benefits in patients with type 2 diabetes. The SUSTAIN-6 trial revealed that semaglutide reduces the risk of major adverse cardiovascular events, including heart attack and stroke, in high-risk patients. Ongoing research is exploring the potential use of semaglutide in treating other conditions, such as non-alcoholic steatohepatitis (NASH) and Alzheimer's disease.ng research. References 2017. Evidenzbasierte und praktische Diabetologie - Unabhängige Fortbildung mit Begeisterung in Jena. Zeitschrift für Allgemeinmedizin, 93(7-8). DOI: 10.3238/zfa.2017.0283-0285 2021. Efficacy and safety of the glucagon-like peptide-1 receptor agonist oral semaglutide in patients with type 2 diabetes mellitus: A systematic review and meta-analysis. Diabetes Research and Clinical Practice, 172. DOI: 10.1016/j.diabres.2021.108656 2024. Adipose Tissue Dysfunction: Clinical Implications and Complications. Contemporary Endocrinology. DOI: 10.1007/978-3-031-72570-8_5 |
| Market Analysis Reports |