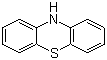
Phenothiazine is an organic compound with a unique structure that consists of a sulfur atom and two nitrogen atoms incorporated into a three-ring system. This compound has been the subject of significant research since its discovery in the late 19th century. Initially synthesized in 1883 by the German chemist August Wilhelm von Hofmann, phenothiazine was discovered as a byproduct of coal tar distillation. Its structure was elucidated later, and it was recognized for its potential biological activity, leading to extensive studies on its applications.
The primary application of phenothiazine lies in the pharmaceutical industry, particularly as an antipsychotic agent. In the 1950s, phenothiazine derivatives were developed as tranquilizers, revolutionizing the treatment of mental disorders. Compounds such as chlorpromazine, which is a phenothiazine derivative, were among the first medications used to treat schizophrenia and other psychotic disorders. These compounds work by blocking dopamine receptors in the brain, which helps to alleviate symptoms such as hallucinations and agitation. The introduction of phenothiazine derivatives marked a significant advancement in psychiatric medicine, leading to more humane treatments for mental health conditions.
In addition to its role in psychiatry, phenothiazine is also used as an antiemetic, helping to prevent nausea and vomiting associated with various medical conditions and treatments, such as chemotherapy. Its efficacy in this area is attributed to its ability to block receptors in the brain that trigger these responses. This dual functionality as both an antipsychotic and an antiemetic has made phenothiazine and its derivatives valuable in clinical settings.
Beyond its medicinal uses, phenothiazine has found applications in other fields, including agriculture. It is utilized as a veterinary drug to treat parasitic infections in livestock and pets. Phenothiazine acts as an anthelmintic, effectively combating worms and other parasites, thereby promoting better health in animals. The use of phenothiazine in veterinary medicine highlights its versatility and importance beyond human health.
Phenothiazine also serves as a dye intermediate in the textile industry. It can be chemically modified to produce various dyes, which are known for their vibrant colors and excellent stability. These dyes are used in a wide range of applications, from fabric dyeing to printing inks. The compound's ability to act as a precursor for dyes demonstrates its significance in industrial chemistry.
Safety considerations are important when handling phenothiazine, as it may pose health risks, including skin irritation and respiratory issues. Proper safety measures, including the use of personal protective equipment and adherence to regulatory guidelines, are crucial to minimize potential hazards.
Recent research has focused on developing new phenothiazine derivatives with enhanced therapeutic properties and reduced side effects. The exploration of novel synthesis routes and modifications aims to improve the efficacy and safety profiles of these compounds, paving the way for future advancements in pharmaceuticals and agriculture.
In summary, phenothiazine is a versatile compound with significant applications in medicine, agriculture, and industrial chemistry. Its discovery has led to groundbreaking treatments for mental health disorders and continues to influence various fields. Ongoing research into its derivatives holds promise for further innovations in health and industry.
References
2022. Medicinal chemistry aspects and synthetic strategies of coumarin as aromatase inhibitors: an overview. Medical oncology (Northwood, London, England), 40(1).
DOI: 10.1007/s12032-022-01916-4
1986. Postsynaptic inhibitory effects of phenothiazines at cholinergic synapses may not involve calmodulin. Naunyn-Schmiedeberg's Archives of Pharmacology, 333(4).
DOI: 10.1007/bf00500024
1991. The discovery of antidepressants: A winding path. Cellular and Molecular Life Sciences, 47(1).
DOI: 10.1007/bf02041242
|





























































 GHS07;GHS08;GHS09 Warning Details
GHS07;GHS08;GHS09 Warning Details