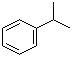
Cumene, also known as isopropylbenzene, is an aromatic hydrocarbon with the chemical formula C9H12. It consists of a benzene ring attached to an isopropyl group (–CH2–CH3) at the para position. This compound is a key intermediate in the production of phenol and acetone, and it is also used as a solvent in various industrial applications.
Cumene was first synthesized in the late 19th century and became commercially significant in the early 20th century due to its widespread use in the petrochemical industry. It is produced primarily through the alkylation of benzene with propylene in the presence of an acidic catalyst, typically using a solid acid catalyst such as alumina or a liquid acid catalyst like sulfuric acid.
The primary industrial application of cumene is in the production of phenol and acetone through the cumene process. In this process, cumene is oxidized to cumene hydroperoxide, which is then cleaved to yield phenol and acetone. The cumene process is one of the most widely used methods for phenol production, with cumene serving as the starting material for these essential chemicals. Phenol is used in the manufacture of plastics, resins, and pharmaceuticals, while acetone is an important solvent and chemical intermediate.
Cumene is also employed as a solvent in the manufacture of resins, paints, varnishes, and adhesives. Due to its solvency properties, it is useful in applications where its ability to dissolve or disperse various substances is required. Additionally, cumene is used as a feedstock for the production of other chemicals, including certain detergents, surfactants, and synthetic lubricants.
Cumene's reactivity and chemical structure make it useful in various chemical processes, particularly in the production of alkylated aromatic compounds and other derivatives. While it is generally regarded as a stable compound, cumene can undergo oxidation or participate in other reactions under specific conditions, which is exploited in industrial processes such as the production of alkylated aromatic compounds.
Due to its widespread industrial use, cumene is also a subject of environmental and health concerns, particularly related to its potential toxicity and flammability. Proper handling and storage are necessary to avoid hazards associated with its volatile nature.
In conclusion, cumene is an important industrial chemical with significant applications in the production of phenol and acetone, as well as in the synthesis of resins and solvents. Its role in the chemical industry, particularly through the cumene process, makes it a vital component in the manufacture of many everyday products.
References
2024. Nano technological approaches for the management of stored insect-pests: present scenario and future prospects. Sustainable Development Goals Towards Environmental Toxicity and Green Chemistry, 13, 145-157.
DOI: 10.1007/978-3-031-77327-3_13
2024. Inactivation of *Escherichia coli* O157:H7 and *Salmonella Typhimurium* on black peppercorns using simultaneous radiofrequency and nonthermal plasma treatment: efficacy and impact on quality. Food and Bioprocess Technology, 17(12), 2545-2554.
DOI: 10.1007/s11947-024-03677-6
2024. Anti-adipogenic activity of oleoresin from *Nigella sativa* L. seeds via modulation of PPAR-γ and C/EBP-α expression in 3T3-L1 adipocytes. Advances in Traditional Medicine, 20(11), 3153-3160.
DOI: 10.1007/s13596-024-00808-4
|















































 GHS02;GHS08;GHS07;GHS09 Danger Details
GHS02;GHS08;GHS07;GHS09 Danger Details