Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Anhui Baishi Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.baishichem.com | |||
 | +86 (552) 492-8807 492-7766 | |||
 | +86 (552) 492-6019 | |||
 | info@baishichem.com | |||
| Chemical manufacturer | ||||
| Jiangsu Yangnong Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yangnongchem.com | |||
 | +86 (514) 8588-8888 8588-9958 | |||
 | +86 (514) 8588-1788 | |||
 | jianghongyan@yangnongchem.com | |||
| Chemical manufacturer since 1999 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon nitrite |
|---|---|
| Name | 3,4-Dichloronitrobenzene |
| Synonyms | 1,2-Dichloro-4-nitrobenzene; 3,4-DCNB |
| Molecular Structure | 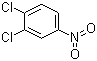 |
| Molecular Formula | C6H3Cl2NO2 |
| Molecular Weight | 192.00 |
| CAS Registry Number | 99-54-7 |
| EC Number | 202-764-2 |
| SMILES | C1=CC(=C(C=C1[N+](=O)[O-])Cl)Cl |
| Melting point | 39-45 °C |
|---|---|
| Boiling point | 255-256 °C |
| Flash point | 123 °C |
| Water solubility | 151 mg/L (20 °C) |
| Hazard Symbols |   GHS07;GHS09 Warning Details GHS07;GHS09 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H319-H411 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P272-P273-P280-P301+P317-P302+P352-P305+P351+P338-P321-P330-P333+P317-P337+P317-P362+P364-P391-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
3,4-Dichloronitrobenzene is an aromatic compound that belongs to the class of chloronitrobenzenes, which are characterized by the presence of both chlorine and nitro functional groups on a benzene ring. This compound was first synthesized in the early 20th century during the exploration of chlorinated aromatic compounds for industrial and chemical applications. Its discovery stemmed from the increasing interest in chloronitrobenzenes as key intermediates in the production of dyes, pesticides, and pharmaceuticals. One of the primary uses of 3,4-Dichloronitrobenzene is as an intermediate in the synthesis of various agrochemicals. The nitro group and chlorine atoms in its structure offer reactivity that is advantageous for creating more complex molecules. It is used in the production of herbicides and insecticides, where its chemical properties help enhance the efficacy of these compounds. Agrochemicals derived from 3,4-Dichloronitrobenzene are particularly valuable in controlling pests and weeds, contributing to improved agricultural productivity. In the dye industry, 3,4-Dichloronitrobenzene serves as a precursor in the synthesis of azo dyes. Azo dyes are widely used in textiles, paints, and printing inks due to their vibrant colors and durability. The chloronitrobenzene framework facilitates the introduction of azo groups, which are responsible for the dye’s chromatic properties. The stability and solubility of these dyes make them popular choices in various commercial applications. Pharmaceutical applications of 3,4-Dichloronitrobenzene are also notable. It is employed as an intermediate in the synthesis of biologically active compounds, including certain types of antibacterial and antifungal agents. Its structure allows for easy derivatization, leading to the development of more potent and selective pharmaceutical agents. Researchers have used this compound in medicinal chemistry to explore new treatments for infectious diseases and other health conditions. The compound has also been used in organic synthesis as a building block for more complex molecules. Its reactivity, particularly due to the electron-withdrawing effects of both the chlorine and nitro groups, makes it a useful reagent in various chemical reactions. This has broadened its application in both academic and industrial research settings, where it is used to develop novel chemical entities. Due to its toxicity, 3,4-Dichloronitrobenzene must be handled with care. It poses risks to human health and the environment, and its use is often regulated in many countries. Despite these concerns, its industrial significance remains strong, as it continues to play a critical role in the manufacture of essential chemicals across various sectors. References 2018. Study of the products of platinum-catalyzed hydrogenation of chlorinated nitro and amino aromatic compounds at high degree of dehalogenation. Russian Chemical Bulletin, 67(8). DOI: 10.1007/s11172-018-2233-1 2021. Continuous hydrogenation of halogenated nitroaromatic compounds in a micropacked bed reactor. Journal of Flow Chemistry, 11(3). DOI: 10.1007/s41981-021-00200-2 1966. Chromatographic analysis of products of catalytic reduction of chloronitrobenzenes to chloroanilines. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 15(10). DOI: 10.1007/bf00848737 |
| Market Analysis Reports |