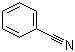
Benzonitrile, with the molecular formula C7H5N, is one of the simplest aromatic nitriles. It consists of a benzene ring directly attached to a nitrile group (–C≡N). Benzonitrile appears as a colorless to pale yellow liquid with an almond-like odor and is known for its stability and versatility in synthetic chemistry.
The discovery of benzonitrile dates back to the 19th century. It was first prepared by Hermann Fehling in 1844 through the dehydration of benzamide using phosphorus pentoxide. This marked an important development in organic synthesis, demonstrating how functional groups on aromatic rings could be manipulated to create new compounds. Subsequent research refined the methods for synthesizing benzonitrile, with current methods often involving the reaction of benzenesulfonyl chloride with sodium cyanide or the ammoxidation of toluene.
Benzonitrile is a key building block in organic chemistry, widely used as an intermediate in the synthesis of pharmaceuticals, agrochemicals, and dyes. The nitrile group can be readily converted into various functional groups, such as amines, amides, or carboxylic acids, making benzonitrile valuable in multiple synthetic pathways. For example, catalytic hydrogenation of benzonitrile produces benzylamine, a compound used in the preparation of numerous pharmaceuticals and polymers.
In the pharmaceutical industry, benzonitrile derivatives are found in drugs that treat cardiovascular conditions, bacterial infections, and neurological disorders. The nitrile moiety often enhances the bioactivity and metabolic stability of these compounds. Additionally, benzonitrile serves as a precursor to the synthesis of herbicides and pesticides, where the nitrile group contributes to the molecule’s ability to disrupt biological processes in targeted organisms.
Benzonitrile also plays a role in materials science, particularly in the production of liquid crystals used in display technologies. Some benzonitrile derivatives exhibit the necessary properties for liquid crystal alignment, contributing to the development of advanced electronic displays.
Another significant application of benzonitrile is in analytical chemistry. It is used as a solvent for infrared (IR) spectroscopy due to its transparency in the IR range and its ability to dissolve a wide range of organic compounds. Furthermore, benzonitrile is utilized in the synthesis of ligands for coordination chemistry, where it helps form stable metal complexes.
Recent astrophysical research has revealed the presence of benzonitrile in space. In 2018, scientists detected benzonitrile in the Taurus Molecular Cloud, marking a breakthrough in understanding the chemistry of interstellar space. This discovery provided evidence for the formation of complex aromatic molecules beyond Earth, offering insights into the origins of organic matter in the universe.
As research continues, the applications of benzonitrile are expected to expand, particularly in pharmaceutical innovation, materials science, and space chemistry. Its simplicity, stability, and functional versatility ensure that benzonitrile remains a valuable compound in both industrial and academic settings.
References
2013. A New Isolate from Fusarium proliferatum (AUF-2) for Efficient Nitrilase Production. Applied Biochemistry and Biotechnology, 171(5).
DOI: 10.1007/s12010-013-0416-7
2020. Design, synthesis, and evaluation of novel 4-amino-2-(4-benzylpiperazin-1-yl)methylbenzonitrile compounds as Zika inhibitors. Bioorganic & Medicinal Chemistry Letters, 30(4).
DOI: 10.1016/j.bmcl.2019.126906
2020. The complete genome sequence of the nitrile biocatalyst Rhodococcus rhodochrous ATCC BAA-870. BMC Genomics, 21(1).
DOI: 10.1186/s12864-019-6405-7
|















































 GHS07 Warning Details
GHS07 Warning Details