Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Ereztech LLC | USA | |||
|---|---|---|---|---|
 | www.ereztech.com | |||
 | +1 (888) 658-1221 | |||
 | sales@ereztech.com | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhengzhou Changkuan Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.changkuantech.com | |||
 | +86 (371) 6376-9919 +86 18530983798 | |||
 | +86 (371) 6360-3986 | |||
 | sales5@changkuantech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Beijing Guofang Dingsheng Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.gfdschem.com | |||
 | +86 (10) 8904-9254 5909-2859 +86 18610472091 +86 18031679351 | |||
 | +86 (10) 5909-2859 | |||
 | gfdskj@163.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | 1,3-Bis(dicyclohexylphosphonium)propane bis(tetrafluoroborate) |
| Molecular Structure | 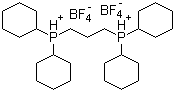 |
| Molecular Formula | C27H50P2.2(BF4).2H |
| Molecular Weight | 612.26 |
| CAS Registry Number | 1002345-50-7 |
| EC Number | 635-571-0 |
| SMILES | [B-](F)(F)(F)F.[B-](F)(F)(F)F.C1CCC(CC1)[PH+](CCC[PH+](C2CCCCC2)C3CCCCC3)C4CCCCC4 |
| Melting point | 154-155 °C* |
|---|---|
| * | Martinelli, Joeseph R. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
1,3-Bis(dicyclohexylphosphonium)propane bis(tetrafluoroborate) is a noteworthy compound in the realm of organic chemistry and catalysis. This compound, also known as a dicationic phosphonium salt, has garnered attention for its role in various catalytic processes, particularly in transition metal-catalyzed reactions. Its discovery and subsequent applications have provided chemists with a versatile tool for enhancing reaction efficiency and selectivity. The compound consists of a propane backbone with two dicyclohexylphosphonium groups attached at the 1 and 3 positions. These positively charged phosphonium groups are balanced by two tetrafluoroborate anions, resulting in a stable salt. The presence of the bulky dicyclohexyl groups imparts significant steric hindrance, which can influence the reactivity and selectivity of the catalytic processes in which it is employed. The discovery of 1,3-Bis(dicyclohexylphosphonium)propane bis(tetrafluoroborate) was driven by the need for highly effective and selective catalysts in organic synthesis. Researchers exploring new avenues in organophosphorus chemistry identified the potential of dicationic phosphonium salts to serve as ligands and stabilizers in various catalytic systems. This led to the synthesis and subsequent investigation of the compound's properties and applications. One of the most significant applications of 1,3-Bis(dicyclohexylphosphonium)propane bis(tetrafluoroborate) is in transition metal-catalyzed cross-coupling reactions. These reactions, essential for forming carbon-carbon and carbon-heteroatom bonds, are foundational in the synthesis of pharmaceuticals, agrochemicals, and complex organic molecules. The compound's ability to stabilize reactive intermediates and enhance the activity of transition metal catalysts has made it a valuable additive in these processes. Additionally, the compound has been employed in the development of novel catalytic systems for olefin metathesis and polymerization reactions. Its unique steric and electronic properties can modulate the behavior of metal catalysts, leading to improved yields and selectivities in these reactions. As a result, it has become an important tool in both academic research and industrial applications. Beyond its use in catalysis, 1,3-Bis(dicyclohexylphosphonium)propane bis(tetrafluoroborate) has also been explored for its potential in materials science. Its ionic nature and the presence of bulky phosphonium groups make it a candidate for the design of ionic liquids and other functional materials with specialized properties, such as enhanced conductivity or thermal stability. In summary, 1,3-Bis(dicyclohexylphosphonium)propane bis(tetrafluoroborate) is a compound of significant interest due to its role in catalysis and potential applications in materials science. Its discovery has enabled the development of more efficient and selective catalytic processes, contributing to advances in organic synthesis and beyond. References none |
| Market Analysis Reports |