Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Austin Chemical Company, Inc. | USA | |||
|---|---|---|---|---|
 | www.austinchemical.com | |||
 | +1 (847) 520-9600 | |||
 | +1 (847) 520-9160 | |||
 | info@austinchemical.com | |||
| Chemical manufacturer since 1976 | ||||
| Scientific Exchange, Inc. | USA | |||
|---|---|---|---|---|
 | www.htscompounds.com | |||
 | +1 (603) 539-7436 | |||
 | +1 (603) 539-7438 | |||
 | sales@htscompounds.com | |||
| Chemical distributor | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Imide |
|---|---|
| Name | 3,3-Dimethylglutarimide |
| Synonyms | 4,4-Dimethyl-2,6-piperidinedione |
| Molecular Structure | 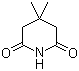 |
| Molecular Formula | C7H11NO2 |
| Molecular Weight | 141.17 |
| CAS Registry Number | 1123-40-6 |
| EC Number | 214-373-4 |
| SMILES | CC1(CC(=O)NC(=O)C1)C |
| Melting point | 145-148 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H335 Details | ||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P272-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P333+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
3,3-Dimethylglutarimide, with the chemical formula C₇H₁₁NO₂, is an organic compound classified as an imide. It is derived from glutarimide, where the two hydrogen atoms on the 3-position of the glutarimide ring are replaced by methyl groups. This modification significantly affects the compound's chemical properties and applications. The discovery of 3,3-dimethylglutarimide is part of the broader study of imides and their derivatives, which began in the early 20th century. The chemical synthesis of 3,3-dimethylglutarimide involves the reaction of glutaric acid with a suitable amine in the presence of a dehydrating agent. This reaction forms the imide structure with the added methyl groups enhancing its stability and reactivity. 3,3-Dimethylglutarimide has found various applications in organic synthesis and industrial chemistry. One of its primary uses is as a reagent in chemical reactions. Due to its imide functionality, it serves as an intermediate in the synthesis of more complex molecules. For example, it can be used in the preparation of various pharmaceuticals and agrochemicals. Its ability to stabilize certain reactive intermediates makes it a valuable component in these synthesis processes. In the pharmaceutical industry, 3,3-dimethylglutarimide is used in the development of certain drugs. It acts as a building block in the synthesis of active pharmaceutical ingredients (APIs) by participating in various chemical transformations. Its role in drug development is crucial for creating compounds with specific therapeutic properties. The compound is also utilized in polymer chemistry. It can be used as a monomer or co-monomer in the synthesis of polymers with specific characteristics. Its incorporation into polymer chains can enhance the material's properties, such as thermal stability and chemical resistance, making it suitable for various industrial applications. Furthermore, 3,3-dimethylglutarimide is used in the production of specialty chemicals. Its reactivity allows it to participate in the synthesis of various fine chemicals and intermediates used in diverse applications, from agrochemicals to advanced materials. Despite its usefulness, handling 3,3-dimethylglutarimide requires appropriate safety measures due to its chemical reactivity. Proper laboratory protocols and protective equipment are essential to ensure safe usage and avoid potential hazards. Overall, the discovery and applications of 3,3-dimethylglutarimide highlight its significance as a versatile reagent in organic synthesis, pharmaceuticals, and polymer chemistry. Its unique chemical properties make it a valuable tool in advancing various chemical processes and industrial applications. References 2006. Synthesis of N-substituted imides via solid-liquid phase-transfer catalytic reaction. Reaction Kinetics, Mechanisms and Catalysis. DOI: 10.1007/s11144-006-0128-x 2010. Complexation of 2,6-Bis(acylamino)pyridines with Dipyridin-2-ylamine and 4,4-Dimethylpiperidine-2,6-dione. The Journal of Physical Chemistry A. DOI: 10.1021/jp1084857 2020. Overview on developed synthesis procedures of coumarin heterocycles. Journal of the Iranian Chemical Society. DOI: 10.1007/s13738-020-01984-1 |
| Market Analysis Reports |