Online Database of Chemicals from Around the World
| Rhodia (shanghai) International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rhodia.com | |||
 | +86 (21) 2408-9317 +86 13601753458 | |||
 | +86 (21) 5483-0035 | |||
 | hank.xue@ap.rhodia.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Interchim Inc. | USA | |||
|---|---|---|---|---|
 | www.interchiminc.com | |||
 | +1 (800) 560-8262 | |||
 | info@interchiminc.com | |||
| Chemical manufacturer | ||||
| Digital Specialty Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.digitalchem.com | |||
 | +1 (603) 563-5060 | |||
 | +1 (603) 563-9288 | |||
 | sales@digitalchem.com | |||
| Chemical manufacturer since 1987 | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | 1,2-Bis(di-tert-butylphosphinomethyl)benzene |
| Synonyms | ditert-butyl-[[2-(ditert-butylphosphanylmethyl)phenyl]methyl]phosphane |
| Molecular Structure | 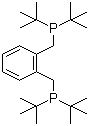 |
| Molecular Formula | C24H44P2 |
| Molecular Weight | 394.55 |
| CAS Registry Number | 121954-50-5 |
| EC Number | 641-386-6 |
| SMILES | CC(C)(C)P(CC1=CC=CC=C1CP(C(C)(C)C)C(C)(C)C)C(C)(C)C |
| Melting point | 59-62 °C |
|---|---|
| Hazard Symbols |    GHS07;GHS08;GHS09 Danger Details GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H317-H319-H335-H372-H411 Details | ||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P271-P272-P273-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P333+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
1,2-Bis(di-tert-butylphosphinomethyl)benzene is a significant compound in organophosphorus chemistry, recognized for its unique structure and broad utility in various chemical processes. This compound features two di-tert-butylphosphinomethyl groups attached to a benzene ring, which contribute to its distinctive steric and electronic properties. The discovery of 1,2-Bis(di-tert-butylphosphinomethyl)benzene stemmed from the need to develop ligands with exceptional steric bulk and electronic tunability. The di-tert-butylphosphinomethyl groups provide both steric hindrance and strong electron-donating effects. The tert-butyl groups are known for their substantial steric bulk, which can stabilize metal centers in catalytic systems and prevent deactivation or precipitation. The phosphinomethyl groups, with their electron-donating properties, enhance the electronic density at the phosphorus atom, making the compound a valuable ligand in metal-catalyzed reactions. In practical applications, 1,2-Bis(di-tert-butylphosphinomethyl)benzene is primarily used as a ligand in transition metal catalysis. The compound's structure allows it to form stable and reactive metal complexes, which are crucial in various catalytic processes. For example, in palladium-catalyzed cross-coupling reactions, the ligand enhances the stability and activity of the palladium catalyst. The steric bulk provided by the di-tert-butyl groups helps to control the reactivity of the metal center, leading to improved selectivity and reduced formation of by-products. Another significant application of 1,2-Bis(di-tert-butylphosphinomethyl)benzene is in olefin metathesis reactions. In these reactions, the ligand facilitates the formation of active metal-carbene complexes, which are essential for efficient olefin metathesis. The compound’s ability to stabilize the metal center while maintaining high reactivity makes it a valuable tool for synthesizing complex organic molecules. The ligand's unique properties also find applications in materials science. The steric and electronic effects imparted by the di-tert-butylphosphinomethyl groups can be utilized to design new materials with specific properties. For instance, the compound has been used in the development of advanced polymers and coordination materials, demonstrating its versatility beyond traditional catalytic applications. Overall, 1,2-Bis(di-tert-butylphosphinomethyl)benzene represents a significant advancement in ligand design. Its distinctive steric and electronic properties enhance the performance of metal-catalyzed reactions and contribute to the development of new materials. The compound's discovery and application highlight ongoing efforts to refine catalytic processes and expand the possibilities of organophosphorus chemistry. References 2005. The methoxycarbonylation of aryl chlorides catalysed by palladium complexes of bis(di-tert-butylphosphinomethyl)benzene. Dalton transactions (Cambridge, England : 2003). DOI: 10.1039/b501868d 2010. Highly selective formation of unsaturated esters or cascade reactions to alpha,omega-diesters by the methoxycarbonylation of alkynes catalysed by palladium complexes of 1,2-bis(ditertbutylphosphinomethyl)benzene. Chemical Science. DOI: 10.1039/c0sc00276c 2017. Tandem Reactions with Renewables. Homogeneous Catalysis with Renewables. DOI: 10.1007/978-3-319-54161-7_7 |
| Market Analysis Reports |