Online Database of Chemicals from Around the World
| Penglai Marine Bio-Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.foodelite.com | |||
 | +86 (535) 598-9265 | |||
 | +86 (535) 598-9263 | |||
 | sales@foodelite.com | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Discovery Fine Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.discofinechem.com | |||
 | +44 (1202) 874-517 | |||
 | +44 (845) 094-4385 | |||
 | pjc@discofinechem.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Nile Chemicals | India | |||
|---|---|---|---|---|
 | www.nilechemicals.com | |||
 | +91 (22) 6631-3162 | |||
 | +91 (22) 2205-4833 | |||
 | sales@nilechemicals.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hubei Seven Eight Nine Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hb789.cn | |||
 | +86 (27) 8811-1833 +86 15802781837 | |||
 | info@hb789.cn niuxiudan@163.com | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Medilink Pharmachem | India | |||
|---|---|---|---|---|
 | www.medilinkpharma.com | |||
 | +91 (79) 3007-0133 | |||
 | +91 (79) 4007-4175 | |||
 | exports@medilinkpharma.com | |||
| Chemical distributor since 1996 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Riverland Trading LLC. | USA | |||
|---|---|---|---|---|
 | www.riverlandtrading.com | |||
 | +1 (336).944-2293 | |||
 | bens@riverlandtrading.com | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2023 | ||||
| Fengzhen Kede Chemicals Tradingco., Ltd. | China | |||
|---|---|---|---|---|
 | www.kedehg.com | |||
 | +86 (0474) 229-4188 | |||
 | +86 (0474) 229-4188 | |||
 | sth@ibojiang.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Jiangsu Raien Environmental Protection Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.raien.cn | |||
 | +86 16651336278 | |||
 | 1780189359@qq.com | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Southern Spechem LLP | India | |||
|---|---|---|---|---|
 | southernspechem.com | |||
 | +91 9391919753 | |||
 | web@southernspechem.com | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Prime Speciality Limited | India | |||
|---|---|---|---|---|
 | primelabs.in | |||
 | +91 9618333911 | |||
 | web@primespl.com | |||
| Chemical manufacturer since 1994 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Tianjin Chemistone Import & Export Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kesiton.com | |||
 | +86 15332170067 | |||
 | martina@chemistone-cn.com | |||
 | QQ Chat | |||
 | WeChat: 008615332170067 | |||
 | WhatsApp:008615332170067 | |||
| Chemical distributor since 1988 | ||||
| chemBlink Standard supplier since 2026 | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Chemos GmbH & Co. KG | Germany | |||
|---|---|---|---|---|
 | www.chemos.de | |||
 | +49 871-966346-0 | |||
 | +49 871-966346-13 | |||
 | chemos@chemos.de | |||
| Chemical distributor | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| ChemPur GmbH | Germany | |||
|---|---|---|---|---|
 | www.chempur.de | |||
 | +49 (721) 933-840 | |||
 | +49 (721) 47-2001 | |||
 | info@chempur.de | |||
| Chemical manufacturer | ||||
| Celtic Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.celticchemicals.co.uk | |||
 | +44 (1656) 749-358 | |||
 | +44 (1656) 746-490 | |||
 | sales@celticchemicals.co.uk | |||
| Chemical manufacturer | ||||
| Classification | API >> Water, electrolyte and acid-base balance regulator >> Electrolyte balance regulator |
|---|---|
| Name | Calcium hydroxide |
| Molecular Structure | 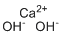 |
| Molecular Formula | Ca(OH)2 |
| Molecular Weight | 74.09 |
| CAS Registry Number | 1305-62-0 |
| EC Number | 215-137-3 |
| SMILES | [OH-].[OH-].[Ca+2] |
| Density | 2.24 |
|---|---|
| Melting point | 580 °C |
| Boiling point | 2850 °C |
| Water solubility | 1.65 g/L (20 °C) |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H318-H335 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P271-P280-P302+P352-P305+P351+P338 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 1759 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Calcium hydroxide, also known as slaked lime or hydrated lime, is an inorganic compound with the chemical formula Ca(OH)₂. It was first described by French chemist Joseph-Louis Gay-Lussac and German chemist Friedrich Wöhler in the early 19th century. Calcium hydroxide is produced by adding water to quicklime (calcium oxide, CaO), a process called slaking. The reaction forms a white powdery substance that is very alkaline. Calcium hydroxide is a white, odorless powder or crystalline solid that is slightly corrosive. It has a melting point of 580°C and is only slightly soluble in water, forming a clear alkaline solution called limewater. The compound has a pH of about 12.4 in saturated solution, making it very alkaline and useful in a variety of chemical processes. Calcium hydroxide is widely used in the construction industry. It is a key ingredient in traditional mortars and plasters. Calcium hydroxide reacts with carbon dioxide in the air to form calcium carbonate, which strengthens the mortar and increases its durability. In civil engineering, it is used to stabilize soils and increase their bearing capacity. The reaction between calcium hydroxide and clay forms more stable compounds, which enhances the performance of the soil in construction projects. In water treatment, it is added to water and wastewater to neutralize acidity and adjust pH to a more neutral level. This helps remove heavy metals and other contaminants from water. Calcium hydroxide is used to remove hardness from water by precipitating calcium and magnesium ions as insoluble compounds, making the water suitable for various industrial processes and domestic use. Calcium hydroxide is used in agriculture to improve soil quality by neutralizing acidic soils, improving nutrient availability, and promoting healthy plant growth. This helps increase crop yields and improve soil structure. It is used as a fungicide and pesticide in organic farming. Its alkalinity helps control certain pests and diseases that affect crops. Calcium hydroxide is used in food processing. It is used as a food additive under E number E526, primarily as a firming agent for canned vegetables and fruits. It helps maintain texture and stability during processing. It is used in the production of lime water and as a base for the preparation of certain foods and beverages, including traditional lime-based beverages. In the chemical industry, calcium hydroxide is used as a reagent in various chemical reactions, including the production of calcium salts and other compounds. It is used in the preparation of precipitates in analytical chemistry, aiding in the separation and analysis of substances. Due to its alkalinity, calcium hydroxide should be handled with care and protective gloves, goggles and dust masks should be worn to prevent skin, eye and respiratory contact. Prolonged contact can cause irritation and burns. It should be stored in a dry, cool place away from moisture and incompatible materials (such as acids). Calcium hydroxide is environmentally safe if used properly. It does not persist in the environment and converts to calcium carbonate over time, which is less harmful. Measures should be taken to prevent large spills and direct discharges into waterways, as high concentrations can affect aquatic ecosystems. References 1987. Root perforations dressed with calcium hydroxide or zinc oxide and eugenol. Journal of Endodontics, 13(8). DOI: 10.1016/s0099-2399(87)80200-5 2024. Antimicrobial and anti-endotoxin activity of N-acetylcysteine, calcium hydroxide and their combination against Enterococcus faecalis, Escherichia coli and lipopolysaccharides. PeerJ, 12. DOI: 10.7717/peerj.18331 2024. Novel combination of nanostructured calcium hydroxide and natural materials: Formulation and characterization. Dental and Medical Problems, 61(5). DOI: 10.17219/dmp/161988 |
| Market Analysis Reports |