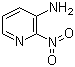
2-Nitro-3-pyridinamine is a versatile organic compound that has attracted much attention for its applications in the synthesis of pharmaceuticals, agrochemicals, and dyes. Its discovery can be traced back to the early exploration of nitroaromatic compounds, a class of chemicals characterized by a nitro (-NO2) group attached to an aromatic ring. The molecular formula of 2-nitro-3-pyridinamine is C5H5N3O2, which features a nitro group attached to a pyridine ring with an amino group at the adjacent position. This unique structure provides the compound with reactive functional groups that are valuable for a variety of chemical reactions.
The discovery of 2-nitro-3-pyridinamine stems from the exploration of nitro derivatives of pyridine, an aromatic heterocycle that is the core structure of many biologically active compounds. Pyridine and its derivatives have been widely studied due to their presence in natural products, vitamins, and pharmaceuticals. Researchers have attempted to introduce functional groups such as nitro and amino groups into the pyridine ring to enhance its reactivity and develop new compounds with potential therapeutic and industrial applications.
One of the main applications of 2-nitro-3-pyridinamine is in drug synthesis. The nitro group can undergo various chemical transformations, such as reduction to amines, and can then participate in further functionalization to produce complex molecules. This makes 2-nitro-3-pyridinamine a valuable intermediate in the production of drugs, especially those targeting bacterial or parasitic infections. Its ability to serve as a building block for heterocyclic compounds makes it a useful precursor for the synthesis of pyridine drugs.
In agrochemicals, 2-nitro-3-pyridinamine has been used in the development of herbicides and insecticides. The nitro group can serve as a precursor for bioactive compounds that interfere with essential biological processes of weeds or pests. Pyridine-based agrochemicals are known for their selective activity against target organisms while minimizing harm to non-target species. This selective toxicity makes pyridine derivatives, including 2-nitro-3-pyridinamine, important ingredients in the development of safer and more effective agrochemicals.
Another important application of 2-nitro-3-pyridinamine is in the dye industry. The nitro group of this compound can undergo substitution reactions to introduce various chromophores, thereby forming colored materials. These dyes are used in textiles, printing inks, and other applications where vibrant and stable coloration is desired. The presence of an amino group on the pyridine ring also provides sites for further modification, allowing the synthesis of a wide variety of azo and nitro dyes with a variety of color properties.
The synthesis of 2-nitro-3-pyridinamine typically involves the nitration of 3-aminopyridine. This process introduces a nitro group at the 2-position of the pyridine ring while maintaining the amino group at the 3-position. The reaction is typically carried out under controlled conditions to prevent over-nitration or unwanted side reactions. Advances in synthetic chemistry have made production methods more efficient, allowing the compound to be synthesized on a large scale for industrial use.
While 2-nitro-3-pyridinamine has many beneficial applications, its safety and environmental impact are also the subject of research. Like other nitroaromatic compounds, 2-nitro-3-pyridinamine can pose a hazard due to its potential toxicity and environmental persistence. Proper handling and disposal procedures must be followed to minimize its impact on human health and the environment. There have been efforts to develop more environmentally friendly synthesis methods and to study the biodegradability of this compound to ensure safer use in industrial applications.
In conclusion, 2-nitro-3-pyridinamine is a valuable compound with wide applications in medicine, agrochemicals and dyes. Its unique chemical structure, with both nitro and amino groups on the pyridine ring, makes it a versatile intermediate for the synthesis of various bioactive compounds and functional materials. Continuous research on its properties and applications may lead to new advances in chemical synthesis and industrial processes.
References
1974. Nucleophilic substitution in 2-nitro-3-halopyridines. Chemistry of Heterocyclic Compounds, 10(6).
DOI: 10.1007/bf00480925
2016. Construction of push-pull systems using beta-formyl-beta-nitroenamine. Russian Chemical Bulletin, 65(9).
DOI: 10.1007/s11172-016-1561-2
2009. Reduction of 2-amino-3- and -5-nitropyridine derivatives with hydrazine hydrate. Russian Journal of Organic Chemistry, 45(1).
DOI: 10.1134/s107042800901014x
|




































































 GHS07 Warning Details
GHS07 Warning Details